Enzyme activity
Equipment list.
- 200 mL beaker
- Fresh chicken liver
- Hydrogen peroxide (6.0% v/v)
- Stop watch
-100 mL measuring cylinder
-
Scissors
- Electronic balance
- Glass rod
- Ice
- Hot plate
- Thermometer
- Ice
Safety - gloves, goggles and labcoats must be worn. Hydrogen peroxide is extremely corrosive.
During this practical investigation we will investigate the impact of temperature on the rate of an enzyme catalyzed reaction;
Catalase is an enzyme found in nearly all living organisms exposed to oxygen. This enzyme catalyses the breakdown of hydrogen peroxide to water and oxygen according to the equation below.
2H2O2 => 2H2O + O2
Hydrogen peroxide is a poison produced during normal cell metabolism and must be broken down before it can do damage to the cell.
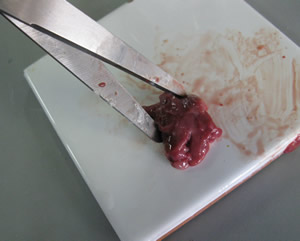
Step 2 - Weigh approximately 3.65 grams of the finely chopped liver, at room temperature.
Step 3 - Add 30 mL of distilled water and 20 mL of hydrogen peroxide solution to a 200 mL beaker and place it on an electronic balance and allow for the solution in the beaker to reach room temperature.


Step 5 - Record the mass loss every 30 seconds.
Using scissors or a glass rod continue to break the bubbles of oxygen so that the gas can escape. Click to see a video.
| time (min) | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 | 3.0 | 3.5 | 4.0 | 4.5 | 5.0 | 5.5 | 6.0 | 6.5 | 7.0 | 7.5 | 8.0 | 8.5 | 9.0 | 9.5 |
| mass loss (gm) |
| time (min) | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 | 3.0 | 3.5 | 4.0 | 4.5 | 5.0 | 5.5 | 6.0 | 6.5 | 7.0 | 7.5 | 8.0 | 8.5 | 9.0 | 9.5 |
| mass loss (gm) |
| time (min) | 0.5 | 1.0 | 1.5 | 2.0 | 2.5 | 3.0 | 3.5 | 4.0 | 4.5 | 5.0 | 5.5 | 6.0 | 6.5 | 7.0 | 7.5 | 8.0 | 8.5 | 9.0 | 9.5 |
| mass loss (gm) |
What is a reasonable hypothesis for this experiment? Choose from the list below.
What must be kept constant in all three investigations?
1) - temperature
2) - volume of solution
3) - mass of liver
4) - size of beaker
5) - size of balance