Some chemical reactions are used to produce electricity for us. These reactions are used as batteries. Atoms exchange electrons very quickly during a chemical reaction. A great deal of electrical energy is used up but not made available to us. By separating the reactants and forcing electrons to travel from one atom to another via an external circuit we can get useful electrical energy from the electrons exchanged.
The situation above is an example of electrons in a chemical reaction used to produce useful work. The electrons are travelling from one electrode to the other via the light globe in the external circuit.
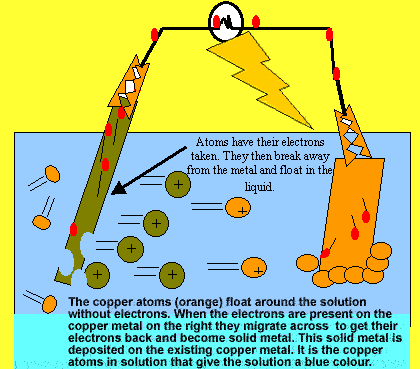
The metal on the left is having its electrons pulled away by the metal on the right. Meanwhile the electrons travel through the external circuit and light up the light globe. This is a simple set up of an electrochemical cell.
Looking at the diagram above explain what happens to the metal on
the left.
The blue solution above is observed to turn clear. Why?
The electrochmical cell (battery)