Water
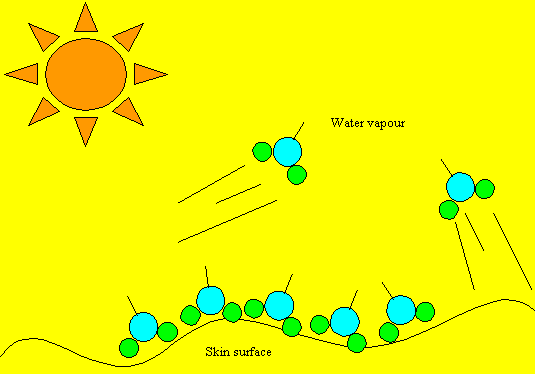
Heat of vapourisation
The heat that each water molecule absorbs in order to turn into a gas is called the latent heat of vaporisation. Big word, but its this that keeps us cool when we perspire. Water molecules on the skin's surface absorb the heat from our body and the sun. When the molecules have absorbed enough heat energy (latent heat of vaporisation) the water molecule flies off into space taking the energy with it. This has the effect of cooling the body .