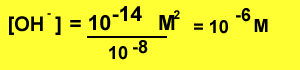pH and Kw
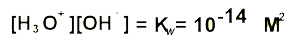
Knowing the hydroxide concentration of a solution we are able to find the pH via the expression above. We can change the expression to solve for the concetration of hydronium ions
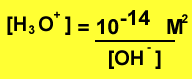
Or we can change the expression to solve for the concentration of hydroxide ions.
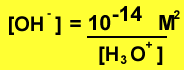
Example. What is the pH of a 0.1 M NaOH solution.

![]()
Example What is the [OH-]
of a solution with pH 8?
Step 1 find the concentration
of hydronium ions ![]()
![]()
Step2 find the [OH-]