Acidity constants of conjugates
Calculating the Ka of conjugates
We know that every acid base reaction is made up of two acid /base pairs. The acid forms a conjugate base while the base forms a conjugate acid. Scan the equation below to see the conjugate pairs.
![]()
As strong acid has a very weak conjugate base and a weak acid produces a strong conjugate base. Take for example the reaction of ethanoic acid with water. The expression for the acidity constant (Ka) is given below.
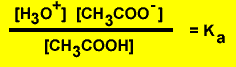
The equilibrium expression for
the reverse reaction is
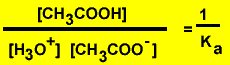
It is obvious
that a very low equilibrium constant for the forward reaction will produce
a very large equilibrium constant for the reverse reaction.