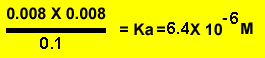Acidity constants
Calculating the Ka
The best way to explain how to calculate the Ka is with an example.
Find the ka of a 0.1M solution of ethanoic acid which is 0.8% ionised.
![]()
Step1 Derive the equilibrium expression.
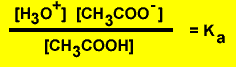
Step 2 Calculate the concentration of each species involved in the expression.
- Since 0.8% is a very low degree of ionisation we can say that the ethanoic acid concentration, at equilibrium, remains 0.8%. Otherwise we would derive it by calculating the amount ionised (0.008 X 0.1) subtracted from the original 0.1 M solution. This gives us 0.0992M close enough to assume 0.1M.
- According to
the above reaction the following expression is true![]()
- Since 0.8% of the ethanoic
acid ionises the concentrations of the hydronium and acetate ions can
be determined.
![]()
- Step 3 calculate
the ka