Acid - Base
Bronsted-Lowry
Substances that
donate protons to other chemical substances are known as Bronsted-Lowry
acids. Substances
that take protons from other chemical substances are known as
Bronsted-Lowry bases. For protons to be transferred an acid and a base
must be present.
The products of an acid/base reaction are also acids and bases known as
conjugate acids and bases. The conjugate acids and bases
will also react with each other to form the original acid and base. The
animation below shows this
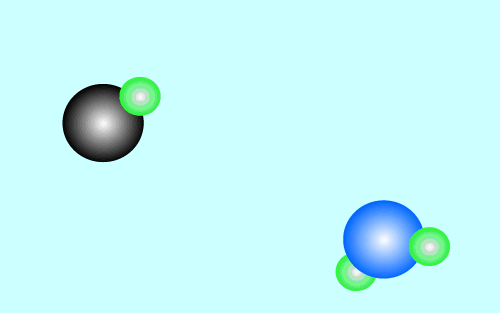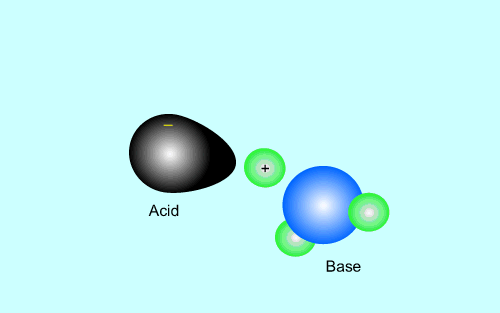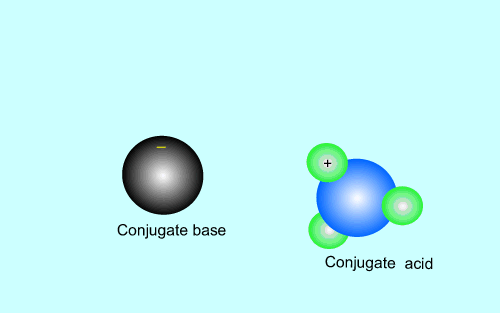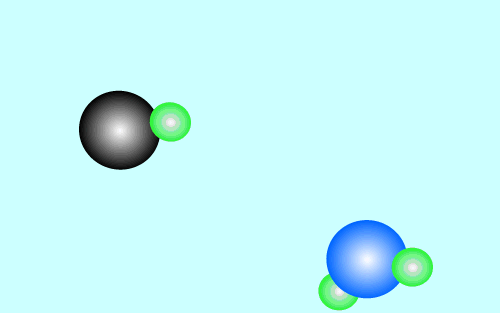
Strong acids have weak conjugate bases while weak acids have strong conjugate bases. This is why reactions that involve weak acids never go to completion, the strong base that is formed quickly reacts with the conjugate acid present to facilitate the reverse reaction. As shown in the animation above.
An acid differs from its conjugate base by one proton(hydrogen ion) and a base differs from its conjugate acid by one proton. Each acid/base reaction involves the transfer of a proton from the acid to the base and two conjugate acid base pairs exist.
For example
![]()
The acid / base
conjugate pair is ![]() /
/ ![]()
The base / acid
conjugate pair is ![]() /
/ ![]()
Notice how the conjugate pairs differ by one proton.
| Acid |
+ |
Base |
Conjugate base |
Conjugate acid |
Equation |
| + |
|
|
|
||
| + |
|
||||
| + |
|
||||
| + |
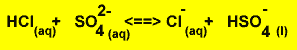 |
||||
| + |
|
||||
| + |
|
||||
| + |