Proton
NMR spectra
The chemical environment of a hydrogen in a molecule is determined by its surrounding atoms.
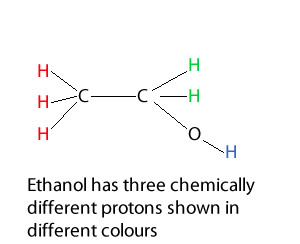
For example, consider ethanol on the right. It has three chemically different protons. The CH3 shown in red the CH2 shown in green and the H bonded to the oxygen, shown in blue.
How many chemically different hydrogens are present in the following molecules and therefore try to predict the NMR spectra produced. Keep in mind the n + 1 rule, where n is the number of neighbouring hydrogens in a different chemical environment. Click to see the molecules.
Butane
NMR

1,2 ethandiol
NMR

Cyclohexane
NMR
