Instrumental analysis
GLC and MS
Investigations in environmental
analysis, drug abuse and forensic investigation of explosive material
often involve the use of GLC and Mass Spectrometry.
Gas liquid chromatography and mass spectrometry each perform very different
tasks but act together to identify a particular substance.
The sample enters the GLC instrumentation first through the injector port
where it is vaporized. An inert gas, then carries the sample through the
column. The chemical compounds in the sample are separated according to
their affinity for the solid stationary phase inside the column. Compounds
with higher affinity for the column material will exit slower than those
with less affinity. The rate at which the different sample components
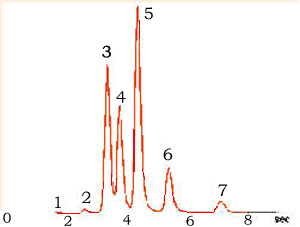
exit the system allows them to be measured and identified. A gas chromatogram
is pictured on the right.
Once separated, the components are passed through the mass spectrum. The compounds now undergo two stages:
First - The separate compounds are ionised by an electron beam. This process causes the compound to break up into into positive fragments. Each compound has a unique way in which it fragments and as such the fragmentation pattern (mass spectrum) can be used to identify each compound.
Second - The fragments enter a magnetic field where they are focused onto
the recorder. The identity and concentration of the particular component
in the sample can be determined. Steroids, stimulants and diuretics are
all substances that can be detected by this gas chromatography-mass spectrometry
method of analysis.
A typical mass spectrum is shown below.

Mass
spectra of banned substances are compared with the mass spectrum of each
compound in the sample in order to identify its constituent compounds.
Probably the most useful information you should be able to obtain from
a MS spectrum is the molecular weight of the compound. This will often
be the heaviest ion observed from the mass spectrum. In the mass spectrum
of pentane, shown above, the heaviest ion has a molecular mass of 142.
This is the molecular mass of the original compound pentane.