2) Take the flask containing the salicylic acid to the a fume cupboard and add 10 mL of acetic anhydride and 1 mL of concentrated sulfuric acid. Swirl and heat on a hot plate at at 50oC until the salicylic acid has dissolved.



4) Set up the Vacuum Filtration apparatus.





From the results shown above
1. Determine the mol of salicylic acid used if the molar mass of aspirin is 180.16 g/mol and the molar mass of salicylic acid is 138.12 g/mol.
Solution
2. Calculate the mass of acetic anhydride from its volume (10 mL) and density (1.08 g/mL)
Solution
3. Calculate the mol of acetic anhydride
Solution
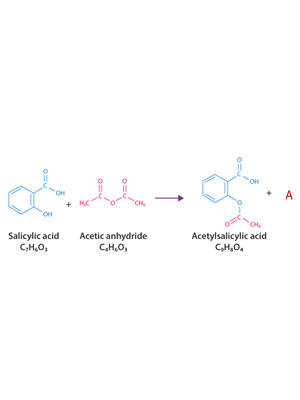
4. On the left is the chemical equation for the formation of aspirin.
i. Which reactant is in excess?
Solution
ii. What is substance A?
Solution
5. Calculate the theoretical yield of aspirin
Solution
6. Calculate the percent (%) yield from the actual product mass .
Solution
i. Is this percentage yield possible? Explain.
ii. Discuss a possible error that would cause this yield.
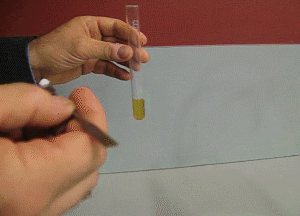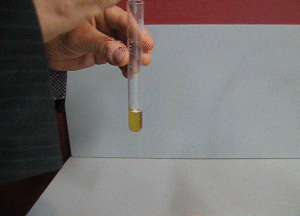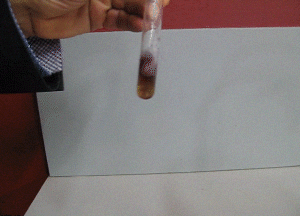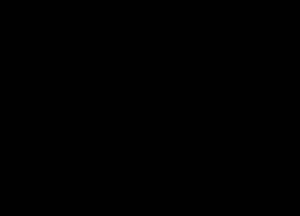
7. FeCl3 turns from a yellow to a purple colour in the presence of salicylic acid, as shown on the right. How can iron(III)chloride be used to check the purity of aspirin formed?
Now do your own synthesis and determine the percentage yield from your own results.