The atom
Filling its energy levels.
Lets see how
the energy levels of different atoms fill.
Look at the animation below. Notice how the total number of electrons
equal the number of protons in the nucleus. A neutral atom has equal
protons and electrons. Note, also, how the lowest energy levels fill
first and once full the atom becomes very stable.
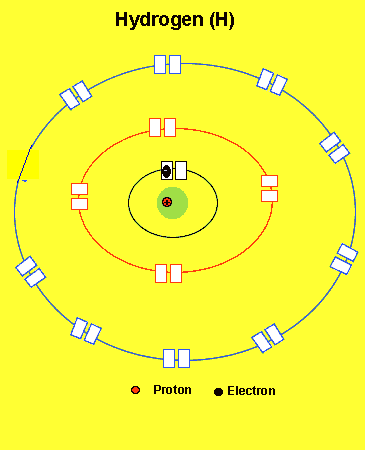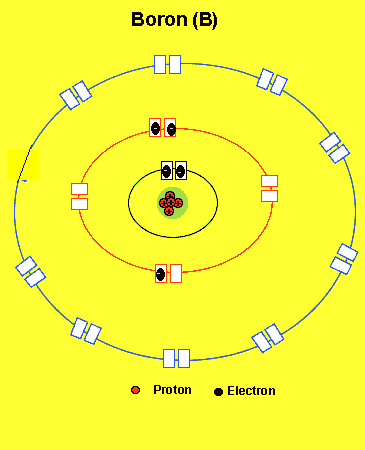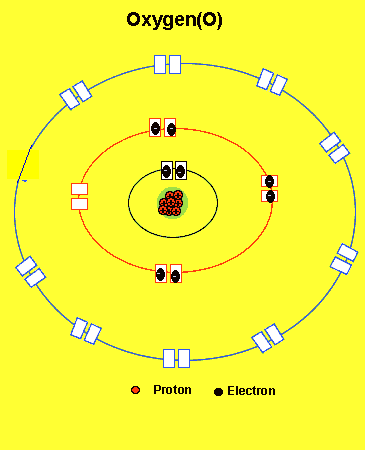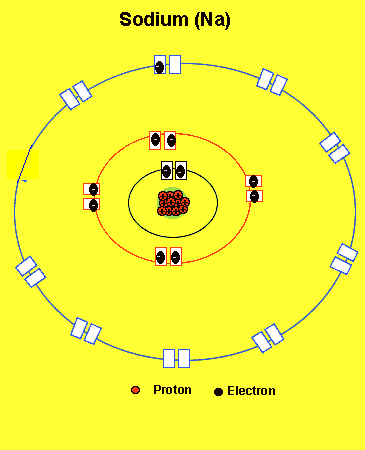
H =1 (one electron in the first energy level)
He =2 (two electrons in the first energy level)
Li = 2,1(two electrons in the first energy level and one in the second level)
Be = 2,2(two electrons in the first energy level and two in the second level)
B =2,3(two electrons in the first energy level and threein the second level)
C =2,4(two electrons in the first energy level and four in the second level)
N =2,5(two electrons in the first energy level and five in the second level)
O =2,6(two electrons in the first energy level and six in the second level)
F =2,7(two electrons in the first energy level and seven in the second level)
Ne =2,8(two electrons in the first energy level and eight in the second level)
Na = 2,8,1(two electrons in the first level, eight in the second and one in the third )