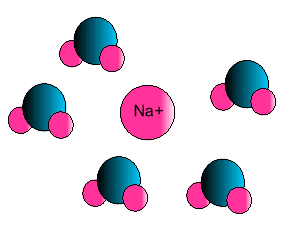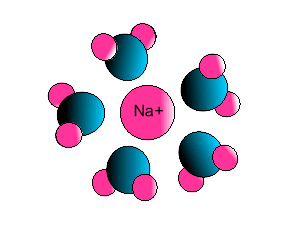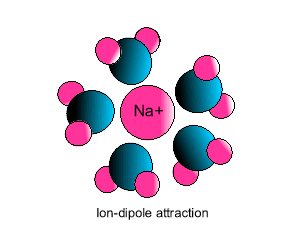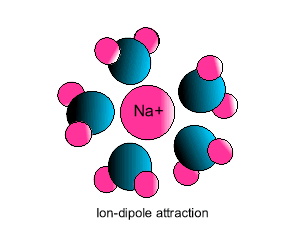Hydrogen bonding
and solubility
|
When an substance
dissolves in water the particles of that substance are separated from
each other by the molecules of water.
Electrostatic attraction between the particles of the substance
and the strong dipoles of the water molecules are responsible for separating the solute particles.
A substance(solute) will dissolve
if the attraction between the particles of the substance and the water(solvent)
molecules is strong enough to overcome the solute-solute and solvent-solvent
forces of attraction. |
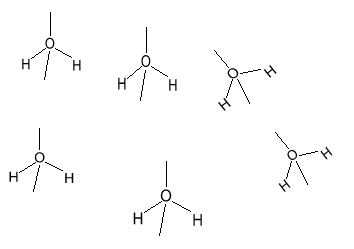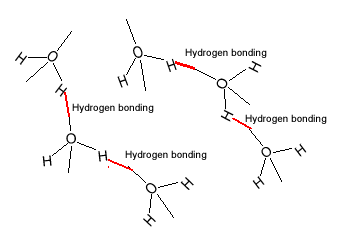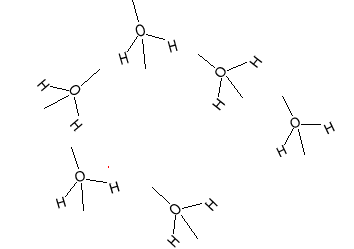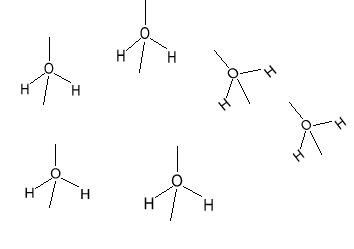
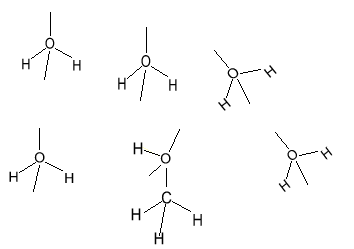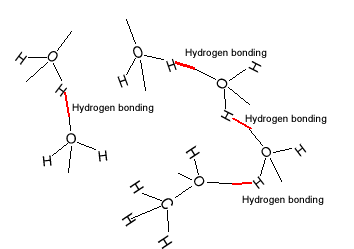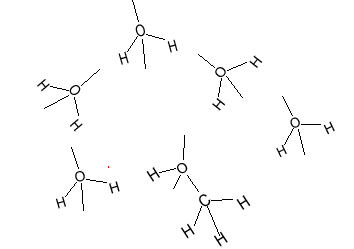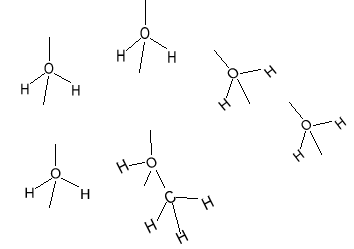
| The water molecule
is polar and hydrogen bonding exists between the molecules of water. As
far as intermolecular bonding forces are concerned hydrogen bonding is relatively
strong. |
|
| Methanol is soluble in water.
Like water, methanol has relatively strong hydrogen bonding acting between
its molecules. The attraction between methanol and water is strong enough
to overcome the methanol-methanol and water-water intermolecular forces
of attraction. |
|
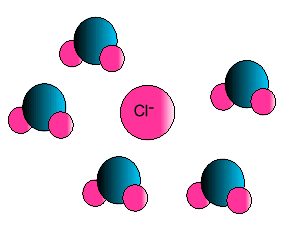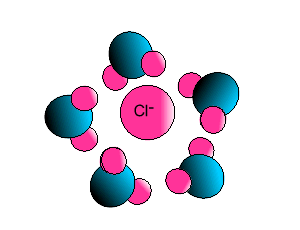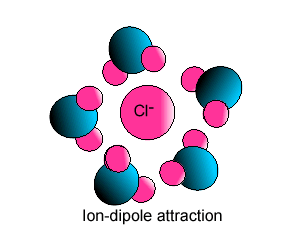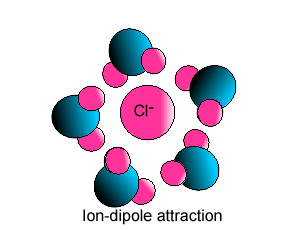
| Some ionic substances, such
as sodium chloride, are soluble in water. The forces of attraction between
the ions and water molecules are strong enough to overcome the ion-ion and
water-water force of attraction. The force of attraction between the ion
and the water molecule is known as ion-dipole attraction. |
|
| Notice how the polar water molecule
binds to the different ions. |
|
|