Ionic bonding.
The giving and the taking.
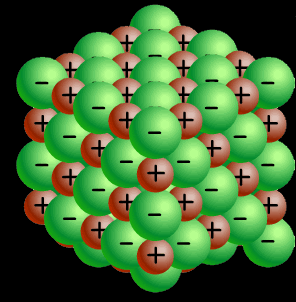
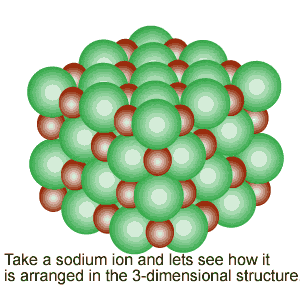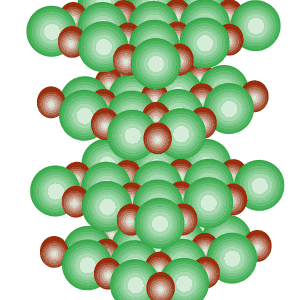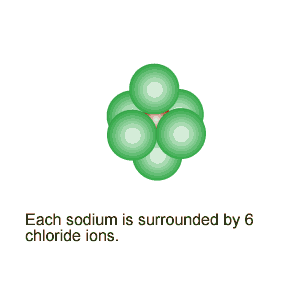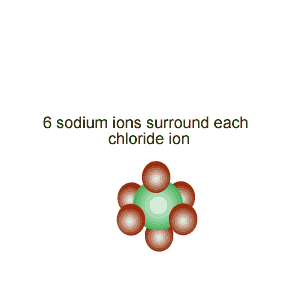
Notice the alignment of the ions and how they create directional, electrostatic forces of attraction to keep the lattice together.
| Ionic bonding. The giving and the taking. |
|
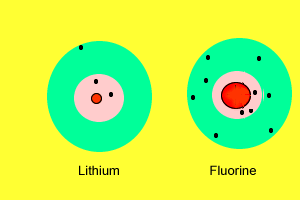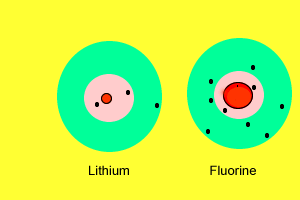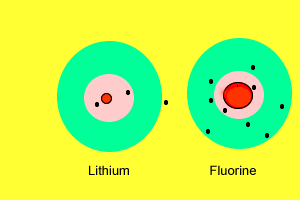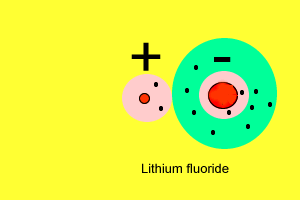
| As we saw before, metals have a very low electronegativity while non-metals have a high electronegativity. When metals react with non-metals electrons are exchanged. Metals give valence electrons to the non-metals. Take the reaction between lithium and fluorine. Lithium has a very low electronegativity and a more stable electron arrangement is desirable. It achieves a more stable arrangement by allowing the electron to be pulled away by the fluorine. The lithium now has a full valence shell (with 2 electrons) while the fluorine also has a full valence shell with 8 electrons. Lithium, which loses an electron, forms a positive ion while fluorine, which gains an electron, forms a negative ion. |
|
 |
The positive and negative ions attract each other to form a 3 dimensional lattice held together by strong, directional, electrostatic forces of attraction between the anions and cations. |
|
The image on the
left shows how the ions of sodium and chlorine are arranged inside the crystal
structure. Notice the alignment of the ions and how they create directional, electrostatic forces of attraction to keep the lattice together. |