Bonding exercises
1) a) Complete the table below.
Molecule |
Symmetry |
| NH3 | |
| CH4 | |
| SO3 | |
| CO2 | |
| C2H6 |
b) Complete the table below.
Molecule |
Intermolecular |
Intramolecular |
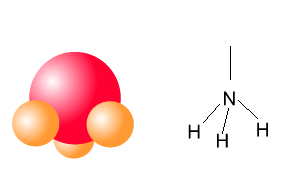 |
||
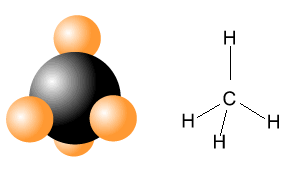 |
||
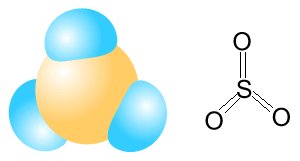 |
||
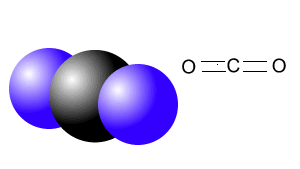 |
||
 |
2) Place the following in order of increasing melting temperature. Give reasons for your choice.
Carbon dioxide, silicon dioxide, diamond, sodium chloride, magnesium oxide, water
Before answering the next question Click to view a summary of bonding and properties of matter.
3) Consider the following, iron (Fe), silicon dioxide (SiO2), magnesium oxide (MgO) and water (H2O).
Which one of the above :
- is a weak conductor of electricity and has the lowest melting temperature;
- can conduct electricity only in the molten state;
- sublimes when heated to very high temperatures;
- melts at high temperatures and conducts electricity in both the molten and solid states.
4) Why does MgO melt at 2,800 oC while NaCl melts at 801 oC and KCl melts at 770 oC ?
a) The reason why MgO has the highest melting point of the three compounds is due to the fact that MgO has:
b) The reason why NaCl has a greater melting point than KCl is best described by the fact that KCl has:
No solutions given for this problem. However in order to answer this question take into account that the melting point is indicative of the strength of the force holding the particles together in the solid. These forces are electrostatic in nature and therefore are directly realated to size of charge and indirectly related to the distance between each charge.
c) Hydrogen gas reacts with oxygen gas to form water according to the following equation.
O2 + 2H2 => 2H2O
a) The intramolecular bonding before the reaction is
The intramolecular bonding before the reaction is pure covalent
b) The intermolecular bonding before the reaction is dispersion forces only
c) The intramolecular bonding after the reaction is polar covalent
d) The intermolecular bonding after the reaction is ihydrogen bonding and dispersion forces.
Hide
b) The intermolecular bonding before the reaction is
c) The intramolecular bonding after the reaction is
d) The intermolecular bonding after the reaction is
Solution