Shapes
exercises
|
Molecule
|
Shape
|
Polar
or non-polar
|
|
|
Triangular
|
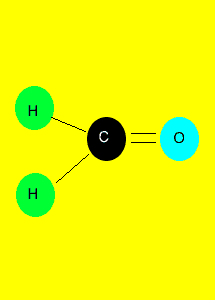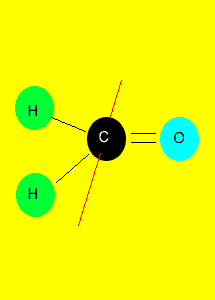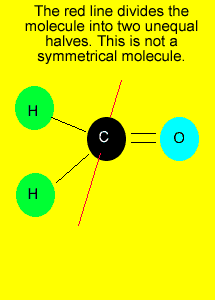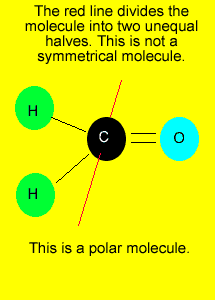 |
Polar
|
|
|
Triangular
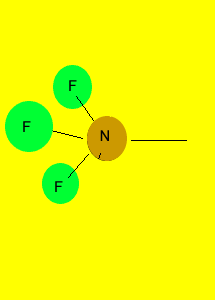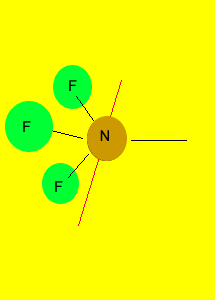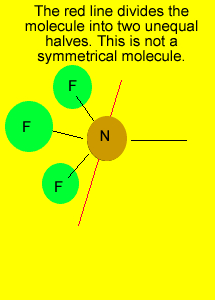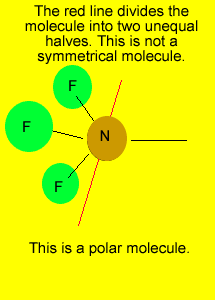
pyramid
|
 |
Polar
|
|
|
"V"
shape
|
 |
Polar
|
|
|
Tetrahedron
|
 |
Polar
|
|
|
"V"
shape
|
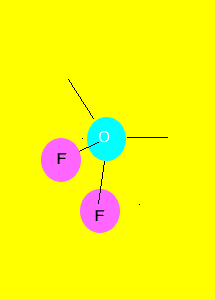 |
Polar
|
|
|
Tetrahedron
|
 |
Non-polar
|
|
|
Linear
|
Non-polar
|
||
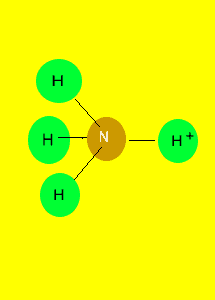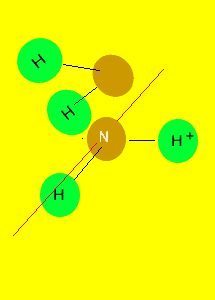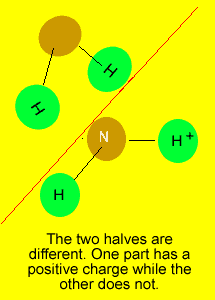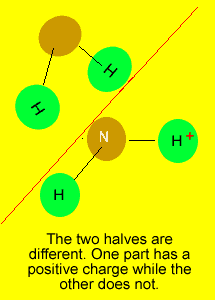
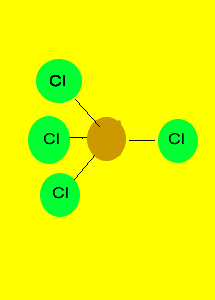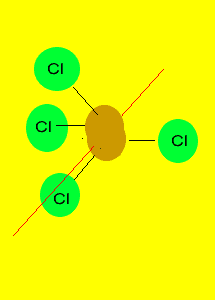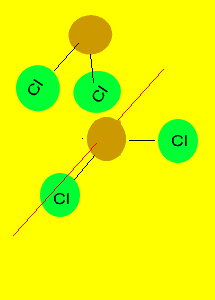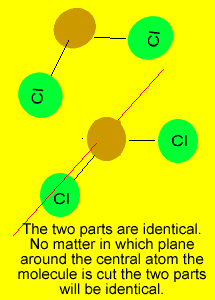
If you can draw
a line through the central atom, in any direction, and in each occasion
divide the molecule into two identical parts, we can say that the molecule
is symmetrical.
The following molecules are symmetrical and it may pay to memorise them.
- O2, N2, Cl2, F2 and any other molecule representing a gaseous element.
- CCl4, CH4, CF4 any other molecule with a central carbon atom bonded to four identical atoms.
- CO2
- Hydrocarbons (molecules made from carbon atoms bonded to hydrogen atoms)
The following molecules are symmetrical and it may pay to memorise them.
- O2, N2, Cl2, F2 and any other molecule representing a gaseous element.
- CCl4, CH4, CF4 any other molecule with a central carbon atom bonded to four identical atoms.
- CO2
- Hydrocarbons (molecules made from carbon atoms bonded to hydrogen atoms)