Metallic bonding-exercises
Consider the lattice structure of Na and Mg. The relative size of both ions in the lattice structure is similar. Explain in terms of electrostatic attraction why the melting point of Na is approximately 98 oC while the melting point of Mg is approximately 648 oC?
Give reasons why aluminium has a melting point of approximately 660 oC while boron has a melting point of close to 2,300oC.
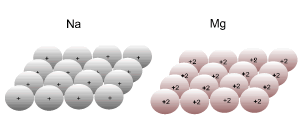
Explain the changes that happen to the metal lattice when it is work hardened.
What is quenching and how does this change the property of the metal?

Consider the diagram on the right of a metal lattice. It shows metals ions with a smaller non-metal atom scattered amongst the ions.
What is the general name given to such a metal?
Explain how the metal's properties change?


Tungsten metal is used in incandescent light globes.
Name two properties of tungsten that make it useful for this purpose.
1)
2)

What name can best be used to describe the metal in the ring?