Properties
of ionic compounds
Property
Reason
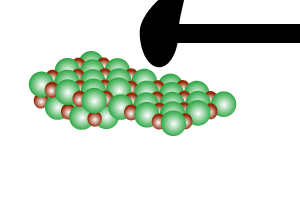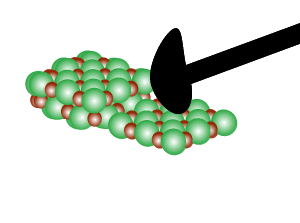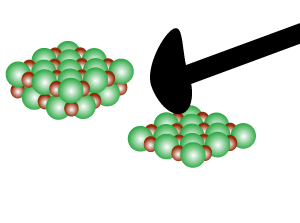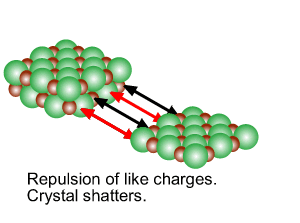
Directional forces
Conducts electricity in both the liquid and aqueous states.
The liquid state is shown on the right. Aqueous state is shown below.
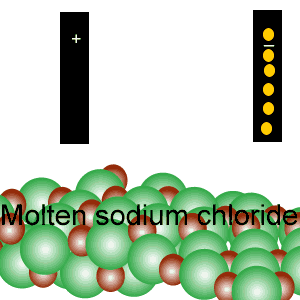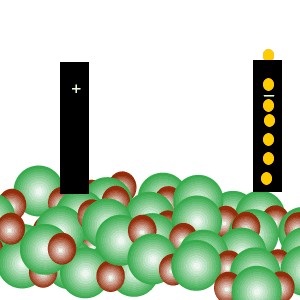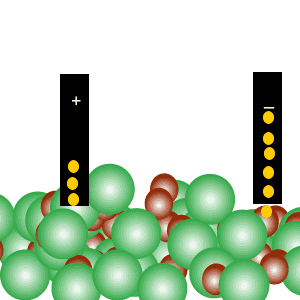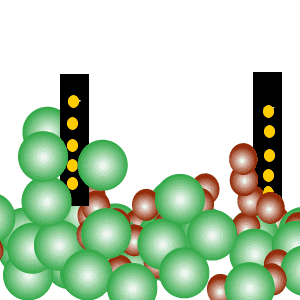
Mobile charges exist is tne liquid state.
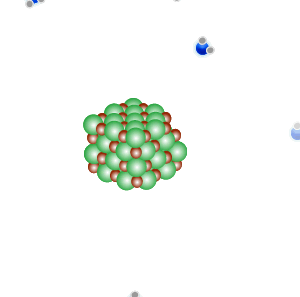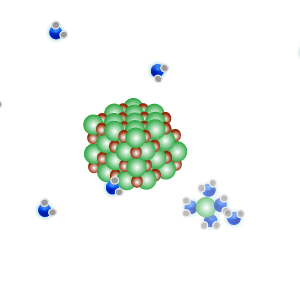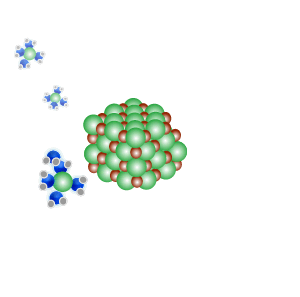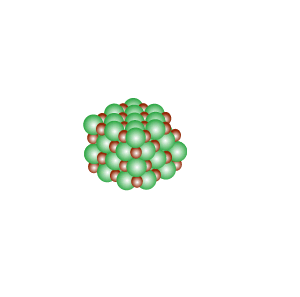
Mobile charges exist when dissolved.