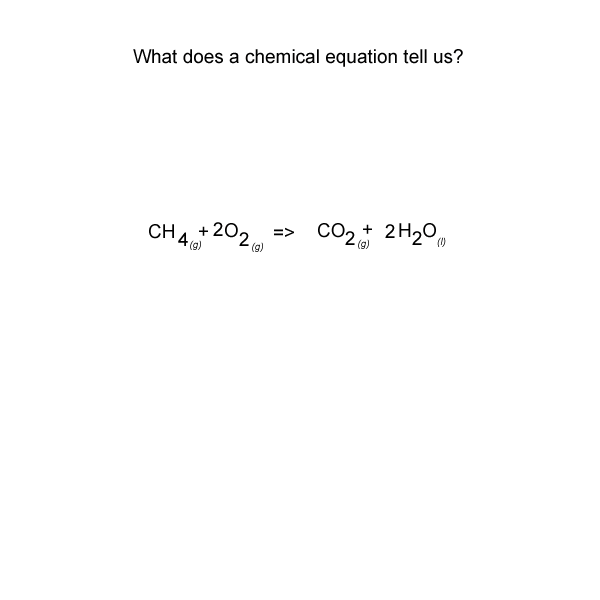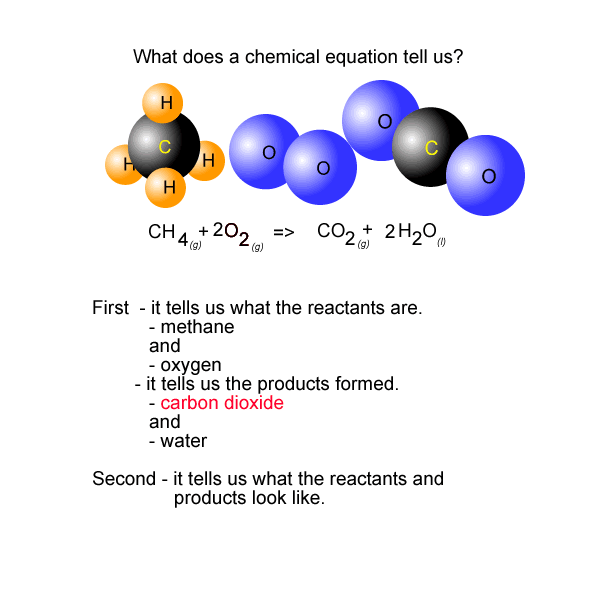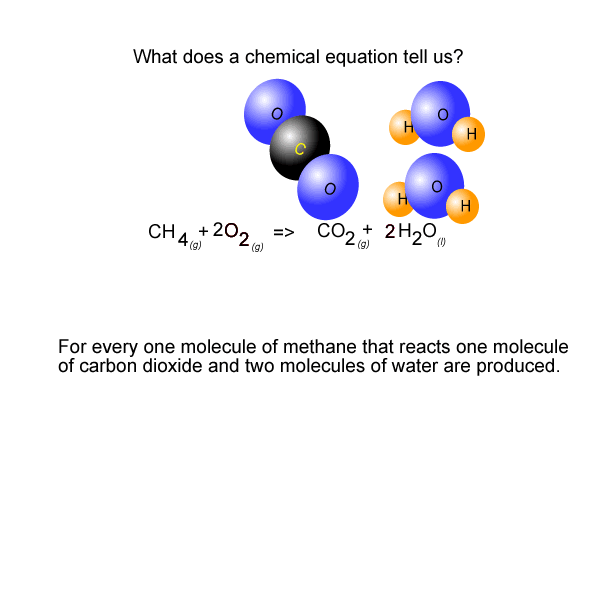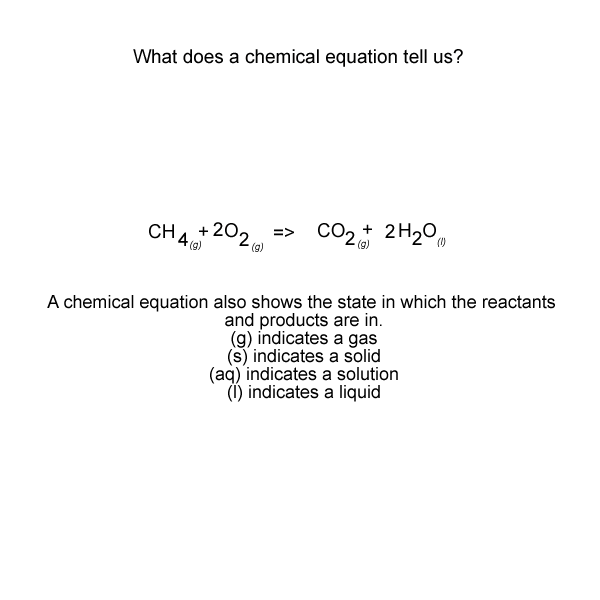
See the video of the animation above.
1) What does the Law of Conservation of Mass state?
2) A balanced chemical equation does not show
3) What must be the same on each side of a balanced chemical equation?
4) Consider the equation below.
Ethene burns in oxygen to produce carbon dioxide and water
according to the equation below.
C2H4(g) + 3O2(g) => 2CO2(g) + 2H2O(l)
a) How many carbon atoms are on the left of the equation?
b) Does this equation obey the Law of Conservation of Mass?
c) Why
d) Each molecule of ethene has
carbon atoms and
atoms of hydrogen.
e) Each molecule of oxygen is made of
atoms of oxygen
f) When 5
molecule/s of ethene react with
molecules of oxygen to form
molecules of carbon dioxide and
of water.
g) The water formed is in the
state.
h) For every two molecules of
ethene that react,
molecules of water form.
i) If 6 molecules of oxygen react, how many molecules of carbon dioxide will form?
5) Consider the equation below.
Sodium carbonate powder is placed in a solution of hydrochloric acid and reacts vigorously to produce carbon dioxide gas, liquid water and dissolved sodium chloride
according to the equation below.
Na2CO3(s) + 2HCl(aq) => CO2(g) + H2O(l) + 2NaCl(aq)
a) How many oxygen atoms are on the left of the equation?
b) Does this equation obey the Law of Conservation of Mass?
. Explain why.
c) Each molecule of carbon dioxide has
carbon atoms and
atoms of oxygen .
d) If 2 molecules of HCl reacted how many molecules of
CO2 are produced?
e)
If 4 molecules of HCl reacted how many molecules of
CO2 are produced?
f) The salt (NaCl) formed is in the
state.
g) For every one molecule of
water that is produced,
molecules of HCl react..
h) If 6 molecules of HCl react, how many molecules of carbon dioxide will form?
6) Consider the chemical equation below. It shows nitrogen and hydrogen gases reacting to form ammonia gas.
N2(g) + H2(g) => NH3(g)
a) All the reactant particles are in the
state
b)
An ammonia molecule consists of
c) Explain why this chemical equation is not considered to be balanced?
d) Select the appropriate prefixes that should be placed in front of each reactant and product in order to balance this equation.
N2(g) +
H2(g) =>
NH3(g)
e) In your balanced equation given in question d) above :
i. how many ammonia molecules appear on the products side of the equation.
ii. how many hydrogen atoms appear on the products side of the equation.