Click here for solution

b) In another experiment, 0.57 × 10–3 mol of 2-chloro-3-methylpentane was analysed in a different mixture under the same conditions.
What would be the expected peak area on the chromatogram associated with this amount of 2-chloro-3-methylpentane?
Click here for solution
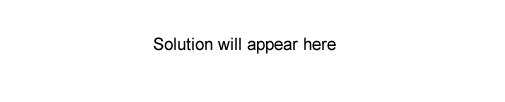
c) The diagram on the right shows the gas chromatogram of a sample containing four straight chain alkanes. The following statements refer to this chromatogram.
I) The boiling points of the compounds arranged from highest to lowest are Z > Y > X > W.
II )The retention times will stay the same if the temperature at which the chromatogram is recorded
is increased, all other conditions remaining constant.
III) Hydrogen gas could have been used as a carrier gas to obtain this chromatogram.
Which of the above statements are true?
A. I only
B. I and II only
C. I and III only
D. II and III only
Click here for solution
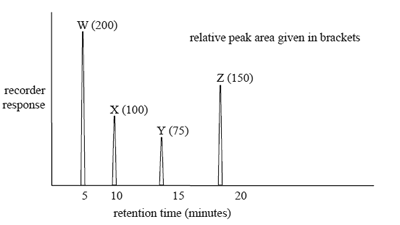
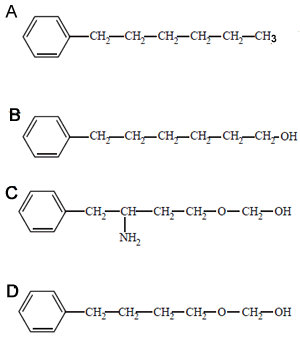
Which of the molecules, shown on the right, will have the greatest retention time on such a reverse phase column?
Solution