Carbon is poured in at the top of the blast furnace along with the iron oxide and calcium carbonate. Air is blown in at the bottom and rises up the blast furnace.
A number of reactions take place.
Click on the blue writing to see the reactions.
First - carbon reacts with oxygen in the air in exothermic reaction to produce carbon dioxide
Second - the limestone decomposes in the heat to produce calcium oxide (quicklime) and carbon dioxide. The limestone undergoes thermal decomposition.
Third -
the carbon dioxide reacts with more carbon to produce carbon monoxide.
Forth - the carbon monoxide removes the oxygen from the iron oxide to produce iron metal and carbon dioxide.
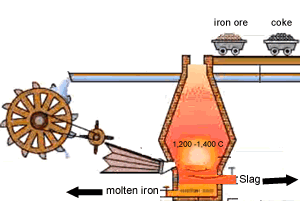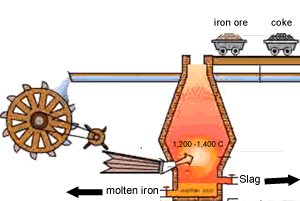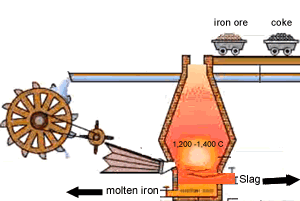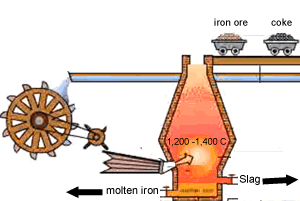
Limestone is calcium carbonate (CaCO3) and it is added to the blast furnace to remove the impurities in the iron ore, as mentioned above. Calcium carbonate is decomposed by heat in the furnace to give calcium oxide (quicklime) and carbon dioxide.
Click for an interactive blast furnace worksheet. Source from ACTIVity
Iron ore often comes buried in sand and rock, commonly known as silica (silicon dioxide). This is the main impurity and is a solid at the furnace temperature, hence we need to remove it before it blocks up the furnace.

Silicon dioxide will react with the calcium oxide to form calcium silicate, known as slag. The equation is given below.
CaO(s) + SiO2(s) => CaSiO3(l)
This slag flows to the bottom of the furnace where it floats on top of the molten iron and is poured off, cooled and used in road construction.
The image on the right shows the slag being poured off.
