| Crystalline structures in plastics |
|
|
|
| Imagine polymers are represented by shoe laces. Sometimes shoe laces are arranged in a neat orderly manner, as shown on the right. When this happens a crystalline polymer is formed. |  |
| Other times there is no order, and the polymer chains just form a big tangled mess, like the shoe laces shown on the right. In this case we say the polymer is amorphous |  |
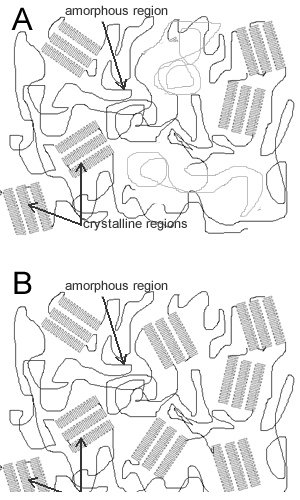
When a polymer grows it doesn't always grow in a linear fashion, the polymer sometimes turns in on itself after it has grown a certain length. This causes a highly ordered structure known as a crystalline structure. The part of the polymer where there is no structure is called the amorphous region. As you can see form the picture on the right, a single polymer chain can be in the crystalline state or in the amorphous state. No polymer is completely crystalline or completely amorphous.
|
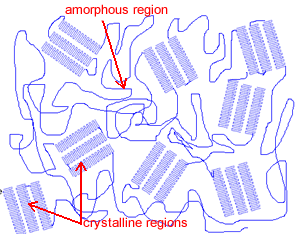 |
Crystallinity makes a material strong, but it also makes it brittle, while amorphous regions create a tough, flexible plastic able to bend without breaking. Depending on the purpose of the plastic it is important to get the percentage of the polymer that is crystalline right. FIbres tend to be highly crystalline polymers. As mentioned before many polymers are a mix of amorphous and crystalline regions, but some are highly crystalline, like fibres and some are highly amorphous, such as polycarbonate. Kevlar is the brand name for a strong plastic, from the nylon family of polymers, that is used to make bullet proof vests. Kevlar is highly crystalline. |
|
| Why does a high degree of crystallinity make the polymer strong? It all comes down to the strength of intermolecular bonds holding the polymers together. The closer the polymer chains can pack the stronger the forces of attraction can be. A high degree of crystallinity gives the polymer a high melting point. | |
| Take a section of a nylon polymer in the crystalline structure, shown on the right. You can see how close the polymer chains come to each other in order for hydrogen bonds to form. | 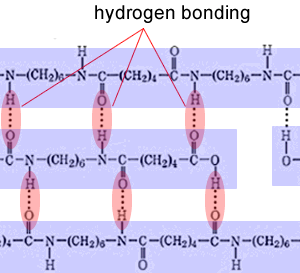 |
1) On the right is a polymer section of Kevlar. Kevlar has an incredibly high tensile strength and isup to five times stronger than steel. It can withstand temperatures of up to 300°C while still retaining its strength. a) What is it about the polymer of Kevlar that allows it to be so strong? b) What can you say about the degree of crystallinity of Kevlar? Explain Solution |
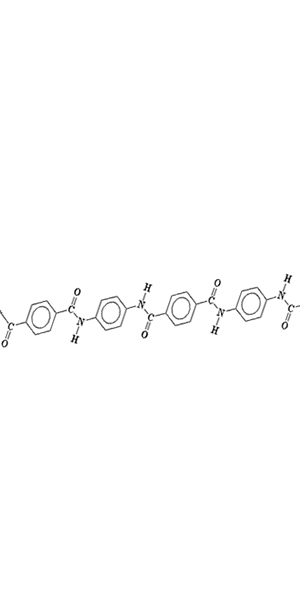 |
2) The image on the right shows the trans and cis configuration of a polymer. Which of the configurations can be used to make the strongest fibre? Explain Solution |
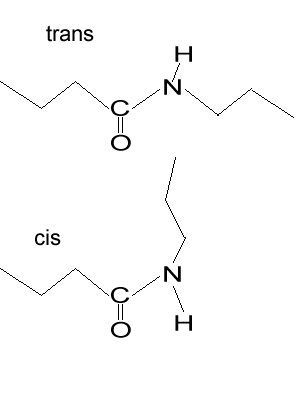 |
3) The image on the right shows two polymers A and B. Which polymer : - has the highest melting temperature, - is most brittle, - has the highest flexibility, - is most likely used to make strong fibres.
4) The image shows amorphous and crystalline regions. Explain the difference between the two regions and explain how they influence the properties of the plastic. |
 |