Ammonia as a fuel

This article was adapted from NewScienctist 3 September 2011 "Ammonia cleans up" Helen Knight.
Along with hydrogen, ammonia could be the answer to developing an emissions-free fuel for cars.
Ammonia produces just nitrogen and water vapour when burnt. Unlike hydrogen, it is relatively easy to store and transport in liquid form and as such will not require costly infrastructure to store it.
Researchers Fleming and Tim Maxwell form Texas Tech University, are developing a system to produce ammonia that can be installed in filling stations. Powered by mains electricity, it first produces hydrogen from water using electrolysis, then combines it with nitrogen from the air to produce ammonia.
To achieve this, the researchers have adapted the Haber-Bosch process used to make ammonia industrially. Their version works on a small scale and can make ammonia fairly cheaply.
In their system, a piston rapidly compresses hydrogen and nitrogen, heating the gases to 400 °C. The mixture is fed into a chamber containing an iron oxide catalyst, which sparks a reaction that further heats the gases and generates ammonia. In a third chamber, the mixture decompresses and cools down to room temperature. As it does so, it pushes against another piston, from which mechanical energy is recovered and fed back to the compressor, significantly cutting the process's power consumption.
Finally, a heat pump cools the mixture down to around -75 °C, liquefying the ammonia for collection.
The team say the whole system could fit within a standard container and could therefore be transported by truck for installation at filling stations, where it could make between 4000 and 40,000 litres of ammonia per day. Maxwell adds that the system has a modular design, so it can easily be scaled up to produce more. The ammonia could be made for just 20 cents per litre, they claim.
Edman Tsang, a chemist at the University of Oxford, says that a mobile unit that can turn water and electricity from renewable energy sources into fuel ammonia would be useful in remote areas. Fleming and Maxwell are already working with the US army and air force, who have each expressed interest in using the technology on the battlefield.
However, Tsang is not convinced that the hydrogen needed to make ammonia can be produced economically using electrolysis. Fleming and Maxwell claim to have got round this hurdle, too.
Conventional cars can already make use of ammonia - they can run on a mixture that is 90 per cent gasoline and 10 per cent ammonia, says Fleming. So-called flexible-fuel vehicles, which use a mixture of gasoline and ethanol, could also be modified to run on a fuel that is up to 85 per cent ammonia. Such vehicles have sensors that constantly detect the exact proportion of gasoline and ethanol being fed through, and adjust their fuel injection and spark timing accordingly. To run on ammonia, the vehicles would need to be reprogrammed and equipped with a fuel tank capable of storing ammonia under pressure.
The team are also designing an engine that could run purely on ammonia. Called the Linear Electric Internal Combustion Engine, it is based on an existing design, known as a free piston engine, in which the burning air and ammonia mixture moves a piston forwards and backwards. This is used to drive a generator, the electricity from which powers a motor that turns the wheels.
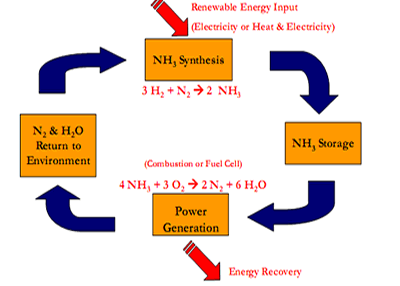
Consider the diagram on the right.
Why is ammonia a non-polluting fuel?
Why is it renewable?
Why is ammonia preferred, as a fuel source, over hydrogen?
At what temperature is ammonia formed? Explain how energy is managed to minimise waste.