Try the following exercises.
Click on the coloured square for more assistance. Double click to hide the information.
|
Exercise
|
Answer
|
| What volume of water should be added to 400ml of a 0.1M NaOH solution in order to change the concentration to 0.075M NaOH. | |
| What volume of water should be added to 80ml of a 0.6M NaOH solution in order to change the concentration to 0.07M NaOH. | |
| 50mls of water are added to 230mls of a 0.1M NaOH solution. What is the concentration of the solution ? | |
|
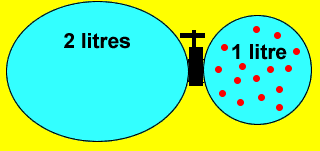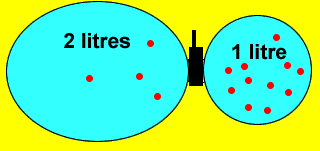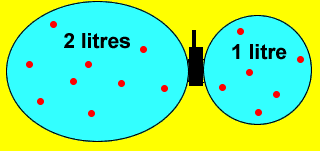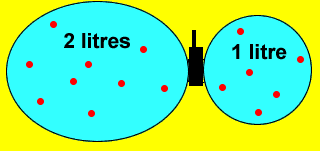
A 1 litre vessel containing 0.5M NaOH solution is connected to a 2 litre vessel containing pure water via a valve (as shown above). When the valve is opened the solute dissolves evenly in both vessels. A sample of water is taken from the 2 litre vessel and analysed. What is the concentration of the sample? |
|
|
A 1 litre NaOH solution is connected to a 6 litre vessel containing pure water via a valve (as shown above). When the valve is opened the solute dissolves evenly through both vessels. A sample of water is taken from the 6 litre vessel and analysed. If the concentration of the sample was found to be 0.45M calculate the concentration of the original solution in the 1 litre vessel? |
|
| What volume
of water should be added to 200ml of a 0.01M NaOH solution
in order to change the concentration to 0.0055M NaOH. solution |
|
| What volume
of water should be added to 400ml of a 0.9M NaOH solution
in order to change the concentration to 0.075M NaOH. solution |