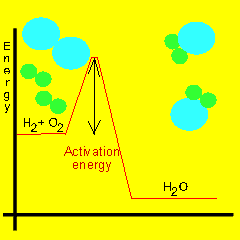
Activation energy
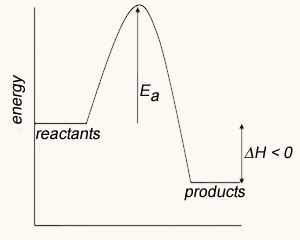
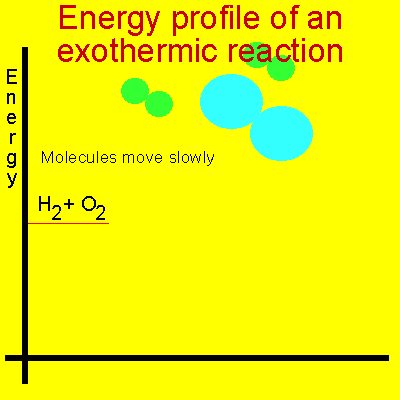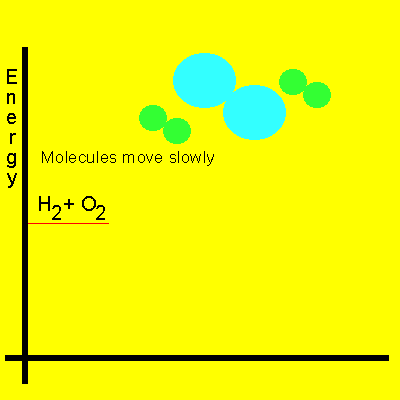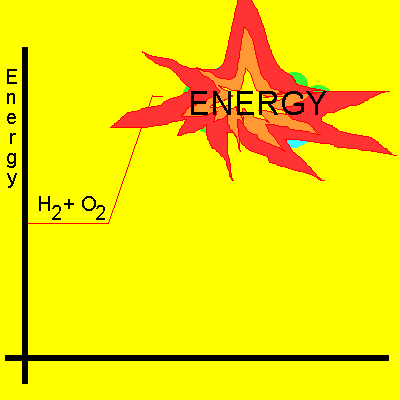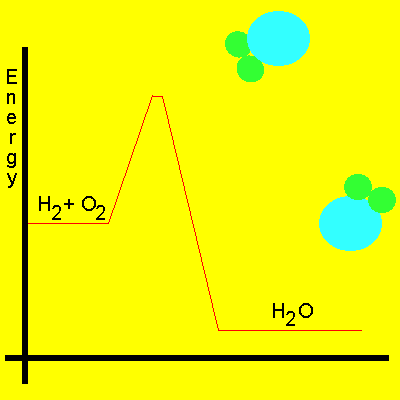
Most reactions involving neutral molecules take place only when they have absorbed enough energy to stretch, bend, or otherwise break one or more bonds. This amount of critical energy is known as the activation energy of the reaction. Energy diagrams, as shown on the left, plot the total energy change to a reaction system as it proceeds from reactants to products.
The activation energies of the forward reactions can be small, large or even zero. A zero activation energy is rare but can occur when a reaction between two oppositely charged gaseous ions takes place. This type of reaction requires next to no activation energy at all as the ions readily attract each other making the reaction spontaneous.
View the video on the right.
In a mixture of methane gas and oxygen gas no collisions occur between the two different types of molecules.
Activation energy is the energy needed to
In an exothermic reaction energy is
In an endothermic reaction energy is

When you open the gas tap natural gas, mainly methane, pours out and mixes with oxygen in the air. But the gas does not burst into flames unless a flame or spark is placed near the gas outlet.
When we use a match to light a fire we provide activation energy to the mixture of gas and oxygen.


In order for a reaction to occur molecules must collide with enough force and in the right spatial orientation to break the chemical bonds that hold them together. Energy, known as activation energy, must be supplied to increase the kinetic energy of the reactant molecules and cause violent collisions. This energy is represented as a small peak on the energy profile diagram shown below.
Take hydrogen and oxygen for example. The energy present at normal room temperatures is not enough to speed up the molecules to any degree. We have to supply a flame or some other form of energy to excite the molecules enough so that they collide with extreme force.
The animation below shows the relative speeds of the molecules prior to the activation energy been supplied. Energy is given off because the product molecules contain less energy than the reactant molecules.
Click to see a FLASH animation of the reaction below.
Keep in mind that even when the energy of collisions between two reactant molecules is greater than that of the activation energy, a reaction may still not occur as the molecules may not collide in the right spatial orientation.
Activation energy does not indicate whether a reaction is spontaneous or not. Most students wrongly assume that a reaction with zero activation energy is spontaneous.
For a reaction to be spontaneous it must occur at the right temperature and:
- be exothermic ( release heat). Another way of saying this is that the forward reaction must have a lower activation energy than the reverse reaction.
-
produce products that have more chaotic motion than the reactants for example the products must be in the gaseous state. Combustion reactions are a perfect example of such reactions, however, even combustion reactions require activation energy.
Activation energy , however, does provide information about the reaction rate. For two similar reactions, under identical conditions, the reaction with the smallest activation energy will occur more rapidly.
Consider the following exothermic reaction, a + b => c + d. If the activation energy for this reaction is given as X what should the activation energy ,given as Y, for the reverse reaction be for the equation to be spontaneous?