Enthalpy of hydration
of copper (II) sulfate
You are provided with :
- a sample of copper sulfate
pentahydrate(CuSO 4.5H2O)
- a sample of anhydrous copper sulfate
(CuSO 4)
- an electronic scales
- 100 mL measuring cylinder
- goggles
- gloves
- labcoat
- a calorimeter
- a thermometer
The aim of this activity is to measure the enthalpy of hydration of copper sulfate using Hess's Law .
We can calculate the ΔH of equation 3 if we know the ΔH of equation 1 and 2.
You will perform this practical in two stages.
First
you will use the secondhand data below to determine the ΔH for reactions 1, below.
Then you will use a calorimeter to measure the ΔH of equation 2.
Click to go to this activity.
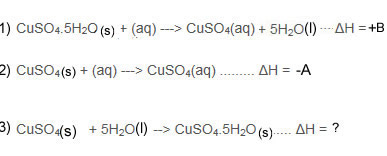
Using Hess's Law you will then work out the ΔH of equation 3.
This has two parts.
Part one - reverse equation 1
Part two - add it to equation 2.






Step 7 Calculate the ΔH. of the reaction below
CuSO4 .5H2O (s) + (aq) => CuSO4(aq) + H2O(l)