In solution, pale yellow-coloured Fe3+(aq) and colourless SCN(aq) form an equilibrium with FeSCN2+(aq). FeSCN2+(aq) is red in colour.
Fe3+(aq) + SCN(aq) => FeSCN2+(aq); ΔH negative
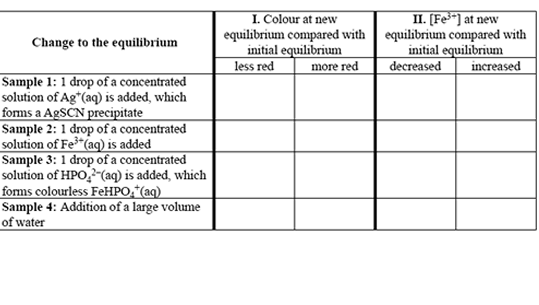
A student investigates this reaction using separate samples of an equilibrium mixture in which significant
quantities of Fe3+, SCN and FeSCN2+ are present. In each case changes are made as indicated in the table
on the right.
Complete the table by placing ticks in the appropriate boxes to indicate the effect of each change on
I. the intensity of the red colour of the solution; and
II. the concentration of Fe3+(aq)
once the new equilibrium has been established.
Solution
The reaction is exothermic. The graph on the right represents the initial concentrations of the ions at equilibrium.
Sketch the changes that would be expected to occur to these concentrations if the temperature of the
equilibrium mixture was increased to a new, constant value.
Solution

Given the following information
Cl2(g) =>2Cl(g); K = 1.13 × 10-6 M at 1100°C
what would be the numerical value of the equilibrium constant for the reaction.
2Cl(g) => Cl2(g) at the same temperature?
A. 8.85 × 105
B. 4.42 × 105
C. 2.26 × 106
D. 1.13 × 106
Solution

Transport of oxygen in the body involves the complex molecules haemoglobin and oxyhaemoglobin.
haemoglobin + oxygen <=> oxyhaemoglobin
If carbon monoxide (CO) is present in the air, poisoning can occur because.
A. the equilibrium constant, K, for the reaction is reduced.
B. CO reacts with oxygen to form CO2, driving the equilibrium to the left.
C. the equilibrium shifts to the left because haemoglobin bonds strongly with CO.
D. CO catalyses the decomposition of oxyhaemoglobin into haemoglobin and oxygen.
Solution
