Equilibrium
The sequence of events
During a reversible chemical reaction in a sealed container the following sequence of events occurs(the sequence is depicted in the image on the right):
- 1. The forward reaction proceeds
and the mass of reactants decreases as the mass of product increases.
- 2. As the concentration of hydrogen and nitrogen decreases
the production of ammonia slows down
- Some molecules of ammonia begin to react together
to form reactant molecules of hydrogen and nitrogen.
- 3. A point is reached where the amount of ammonia formed
equals the amount of ammonia broken down. The amount of ammonia present has reached a point where its constant, however, that does not mean that the reaction has stopped. At this point the rate of the froward reaction, ammonia synthesis, equals the rate of the reverse reaction, ammonia breakdown. This is known as the dynamic equilibrium point. The word "dynamic" is used and refers to the fact that molecules are still colliding to initiate a chemical reaction and the forward rate of reaction is equal to the rate of the reverse reaction so that there is no observable net change in amount of product formed or amount of reactant remaining.
Two conditions must be met for a dynamic equilibrium to be achieved.
1- The reaction must be reversible
2 - The system must be a closed system. That is, there is no exchange of matter with the surroundings just energy.
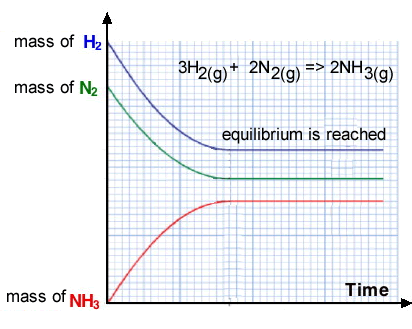
As you can see from the mass vs time graph above, during equilibrium :
- the amount of each reactant
and product stays the same. If the reactants and products were gases then
the total pressure would remain constant;
- the reaction is incomplete;
- the temperature is constant.
Take the formation of HCl from H2 and Cl2 gases as shown below
Cl2(g) + H2(g) ![]() 2HCl(g)
2HCl(g)
In a sealed container the reaction reaches equilibrium. That is, the rate of the forward and backward reactions are equal. At this point we still have some unreacted Cl2(g) and H2(g)
In order to react all the Cl2(g) and H2(g) we need to constantly remove a product (HCl). Removing HCl takes away the reactant for the backward reaction and slows or prohibits the backward reaction from taking place. This prevents the formation of Cl2(g) and H2(g) via the backward reaction and maximises the yield of HCl.