Equilibrium
Le Chatelier's Principle
Exercises -Solutions
Le Chatelier's Principle
Exercises -Solutions
![]()
The above reaction takes place
in a 4 litre sealed container and allowed to proceed to equilibrium. The
table below indicates the changes that occur to the equilibrim mixture.
Fill in the table and suggest how the equilibrium will shift to oppose
the changes.
| Change |
Mass
of methane |
Mass
of water |
Mass
of carbon dioxide |
Equilibrium
shift |
| Decrease
in volume |
net
increase |
net
increase |
net
decrease |
proceeds
to the left |
| Increase
in pressure by the addition of helium gas |
no
change |
no
change |
no
change |
no
change |
| Increase
in temperature |
net
decrease |
net
decrease |
net
increase |
proceeds
to the right |
| Addition
of hydrogen gas |
net
increase |
net
increase |
net
decrease |
proceeds
to the left |
| Increase
in volume |
net
decrease |
net
decrease |
net
increase |
proceeds
to the right |
| Removal
of hydrogen |
net
decrease |
net
decrease |
net
increase |
proceeds
to the right |
| Decrease
in temperature |
net
increase |
net
increase |
net
decrease |
proceeds
to the left |
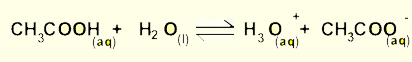
The above reaction takes place in a 1litre sealed container and allowed to proceed to equilibrium. The table below indicates the changes that occur to the equilibrim mixture. Fill in the table and suggest how the equilibrium will shift to oppose the changes.
|
Change
|
Mass
of acetic acid
|
Mass
acetate ions
|
Mass
of hydronium ion
|
pH
|
Equilibrium
shift
|
|
Addition
of 2 lires of water
|
decrease
|
increase
|
increase
|
decrease
|
proceeds
to the right
|
|
Water
is allowed to evaporate
|
increase
|
decrease
|
decrease
|
increase
|
proceeds
to the left
|
|
Addition
of NaOH
|
decrease
|
increase
|
Overall
decrease but the reaction will favour its production
|
Overall
increase because of the OH ions added
|
proceeds
to the right
|