Calculating
"Kc"
Example 3
2NH3(g) + 3O2(g) ![]() 2N2(g) + 6H2O(g)
2N2(g) + 6H2O(g)
Three moles of ammonia and four moles of oxygen gas were mixed in a 2 litre sealed vessel.The reactants reacted according to the equation above. The system was allowed to reach equilibrium , at which point the mass of nitrogen was measured at 56.0 grams. Calculate the equilibrium constant.
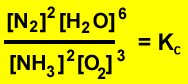
Firstly calculate the mole of nitrogen gas formed. 56 / 28 = 2 mole of Nitrogen gas
For every one mole of nitrogen gas produced one mole of ammonia is used. So 2 mole of ammonia reacts. This leaves 1 mole of ammonia present at equilibrium.
Secondly calculate the mole of oxygen that remains at equilibrium. Following the same procedure as above we find that 3 ((3/2) X 2) mole of oxygen gas reacts. This leaves 1 mole of oxygen at equilibrium.
Thirdly calculate the mole of water formed. The mole of water formed is three times the mole of nitrogen formed. Six mole of water is present at equilibrium.
To calcualte the concentration of each species divide the number of mole present by the volume of the vessel.
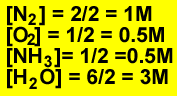
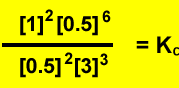
![]()