Esters
| Esters have many uses in nature and in industry. | |
| Fat is stored in our body as an ester. Fatty acids are chemically bonded to an alcohol called glycerol to form a triglyceride. This way fat is stored in our fat cells. |
|
| Esters are used as organic solvents. One use is in the preparation of solutions of organic metal complex dyes which exhibit brilliant shades and excellent properties. These dyes are used in the production of metalized polyester film and metal foil lacquers. |
|
The fragrance in perfumes is often due to the presence of esters. |
|
| The smell of flowers is also due to the presence of a particular ester or a mixtrue of several esters. Jasmine, for example contains the ester benzyl acetate. |
|
The structural formula of benzyl acetate is shown on the right.
|
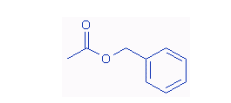 |
| Benzyl acetate is just one of many esters used as flavours in the food industry. It imparts fruit flavors like those of banana, strawberry, pear and apple and is thus used in the food industry. |  |
Esters are chemical compounds responsible for the fruity smells present in processed food. Many natural flavours and smells are due to the presence of esters in flowers and fruits. An ester is formed from the reaction between an alcohol and an acid. Its name is given by adjusting the names of the alcohol and acid that formed the ester. For example
When methanol(alcohol) reacts with ethanoic acid the ester methyl ethanoate is formed. The name is derived by placing the name of the alcohol first. The anol part of the alcohol is replaced with yl to form the word methyl. The ic part of the acid is replaced with ate to form the name ethanoate. Putting the two words together gives us the name of the ester, methyl ethanoate.
|
Alcohol
|
Acid
|
Ester
name
|
|
ethanol
|
ethanoic
acid
|
|
|
pentanol
|
ethanoic
acid
|
|
|
butanol
|
ethanoic
acid
|
|
|
methanol
|
ethanoic
acid
|
|
|
propanol
|
ethanoic
acid
|
|
|
methanol
|
salycilic
acid
|
|
|
ethanol
|
butyric
acid
|
|
|
pentanol
|
butyric
acid
|
|
|
pentanol
|
salycilic
acid
|
|
Some esters
you may recognise by their smell are:
Ethyl ethanoate = Apple
Ethyl butanoate = Pineapple
Octyl ethanoate = Orange
Butyl methanoate = Raspberry
Pentyl butanoate = Banana
Ethyl methanoate = Rum
Methyl salicylate = Oil of wintergreen
Research the esters above and give their uses society or industry.
Complete the table below
|
Ester
|
Alcohol
|
Organic
acid
|
| Ethyl ethanoate |
Ethanol
|
Ethanoic
acid
|
| Ethyl butanoate |
|
|
| Octyl ethanoate |
|
|
| Butyl methanoate |
|
|
| Pentyl butanoate |
|
|
| Ethyl methanoate |
|
|
| Methyl salicylate |
|
|
Visit your local food outlet and identify the esters in:
-nail polish
remover
-Dencorub or DeepHeat (these are brand names)
-icecream
-flavoured drinks
-butter
-confectionary.
Senior chemistry
Why is it necessary to use numbers, such as, 3-octyl methanoate? Draw the structure.
Draw the isomers of octanol.
How many possible esters can be formed by reacting an octanol with methanoic acid? Draw them.
Name the ester formed from ethanoic acid and butan-2-ol
Esters have small molecular weight and very nice odor. They are responsible for the pleasant odor of jasmine, roses, etc. and for the odors of apples, bananas, strawberries, etc. The higher the molecular weight, the weaker the odors they carry are.
Unsaturated esters have stronger odour than the saturated ones.
1) Why do esters with higher molecular weight have a weaker fragrance?
2) What is meant by the term saturated and unsaturated?



