The
ideal gas equation
PV =nRT
Exercises
PV =nRT
Exercises
1) 1.00 litre
of a gas has a mass of 1.63g at 327oC and 104kPa. Determine
the molecular mass of this gas.
![]() Roll the cursor over the button to see the answer.
Roll the cursor over the button to see the answer.
2) What is the
density of a pure sample of CO2 placed in a closed container
at 27oC and 93.3kPa?
Atomic mass C=12,O=16.
Atomic mass C=12,O=16.
![]() Roll the cursor over the button
to see the answer.
Roll the cursor over the button
to see the answer.
Atomic mass N=14.
![]() Roll the cursor over the button
to see the answer.
Roll the cursor over the button
to see the answer.
Atomic mass O=16.
![]() Roll the cursor over the button
to see the answer.
Roll the cursor over the button
to see the answer.
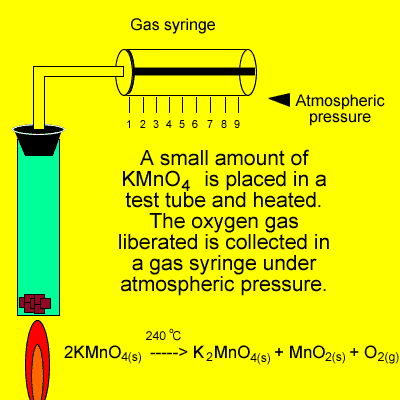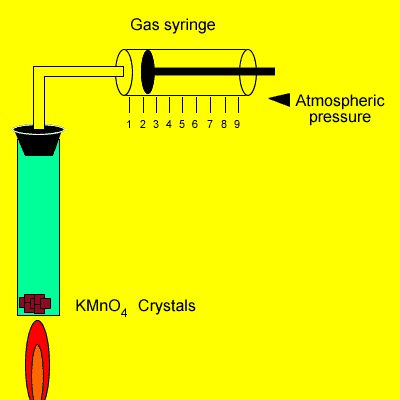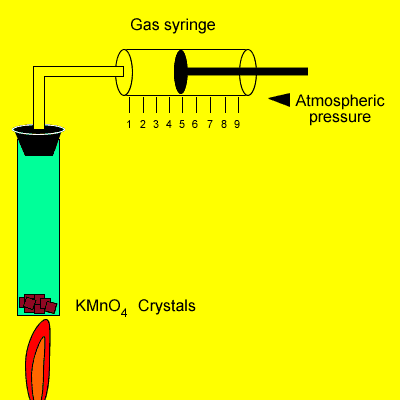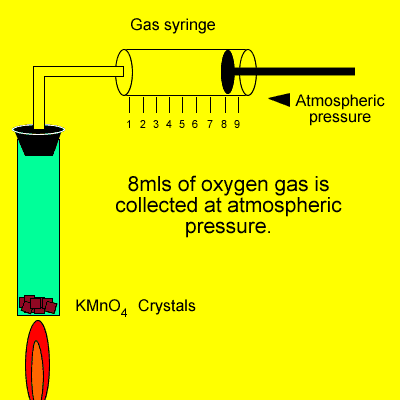
An experiment was conducted
in class where oxygen gas was produced as shown on the right. If the
temperature is 240oC what mass of oxygen gas is collected
during this experiment?
Ask you teacher for assistance.
Continue
with miscellaneous gas exercises