Oxygen 16O and paleoclimatology
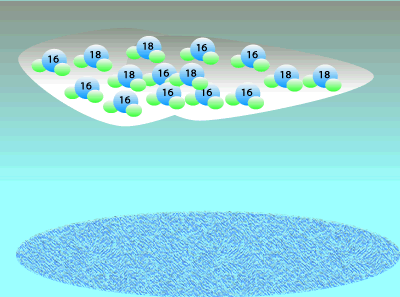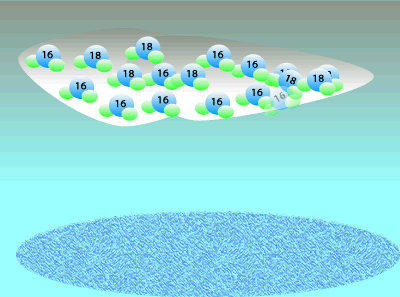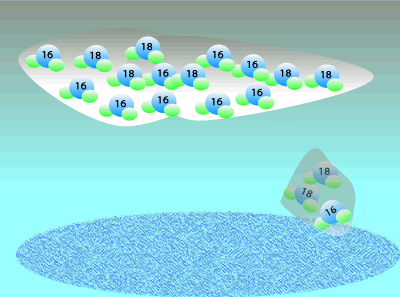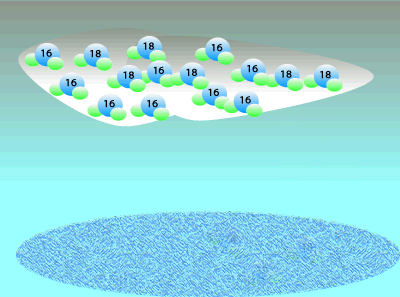
Oxygen consists of three isotopes: 16O, 17O, and 16O. A large percentage ((99.762%) of the Earth's oxygen is 16O and the rest we can say is 18O. A tiny, negligible, amount is 17O but we will ignore this for simplicity. Although the percentage of 18O is small we can still measure the ratio of 18O to 16O. This means that any mineral containing oxygen, or ice, will have a measurable ratio of 18O to 16O atoms. This ratio of the 18O to 16O is temperature dependent so we can determine the temperature at the time by simply measuring the ratio of these two isotopes.

When water vapour condenses to form rain it is the heavier, less mobile water molecules that form rain.
This leaves a greater percentage of the lighter water molecule in the air as water vapour.

1) What is an isotope?
2) What do isotopes have in common?
3) Two water molecules are at the same temperature, they have the same average kinetic energy. One has an 16O isotope while the other has an 18O isotope. Which one travels with greater speed? Explain.
4) In order to evaporate water molecules must break forces of attraction that act between water molecules in the liquid state. Fast moving molecules break their bonds easier than slow moving molecules. Explain why clouds are enriched with 16O isotope.
5) Condensation occurs when water molecules slow down so that forces of attraction take hold and bind water molecules together in the liquid state. Explain why rain is enriched with 18O isotope.
6) Explain how glaciers have a greater percentage of 16O than sea water found in the equator.
7) How can the temperature of a region that prevailed millions of years ago be estimated using oxygen isotopes?

We can therefore use oxygen isotope ratio from fossils as well as from glacial ice to work out how global climate has changed over millions of years.
We can thus make a few generalisations when looking at the information aobve.
1. Greater percentage of 18O isotopes are present in a mineral as the temperature in the area the mineral is formed gets lower.
2. The more glaciers that are formed the greater the percentage of 18O present in the oceans and so the greater the percentage of 18O in marine minerals.
3. The percentage of 18O decreases in glacial ice as the temperature also decreases.
What impact would a colder world temperature have on the percentage of 18O in fossil deposits?