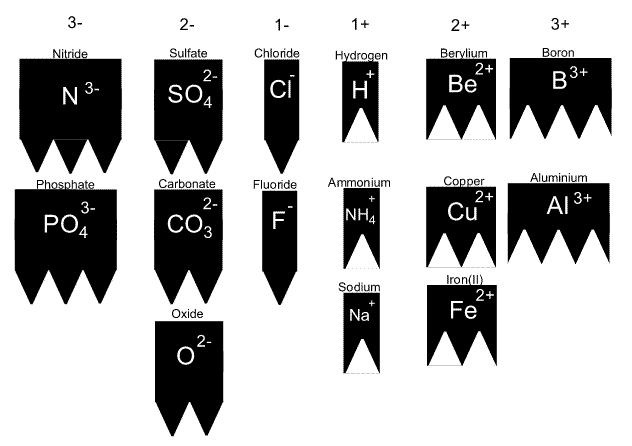
For example
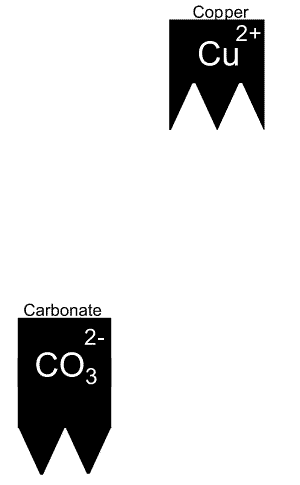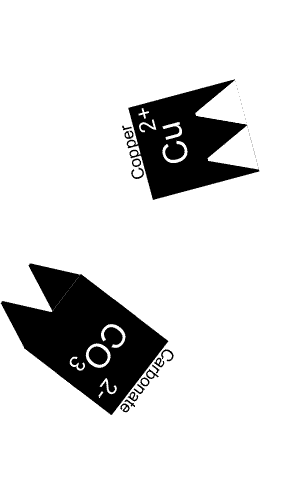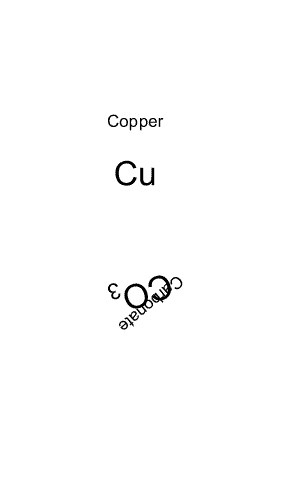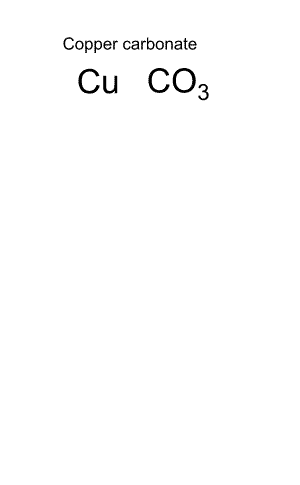
Give the name and formula of the compound formed when copper and carbonate react.
The solution is animated on the right.
We combine the ions until they are perfectly matched. In this case one of each ion is all it takes. The formula is therefore given as CUCO3.
To name the compound we always put the positive ion first.
Give the name and formula of the compound formed when aluminium and carbonate react.
The solution is animated on the right.
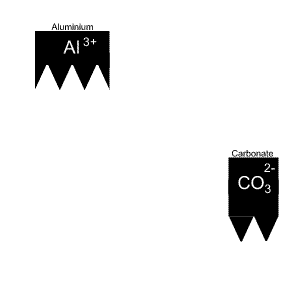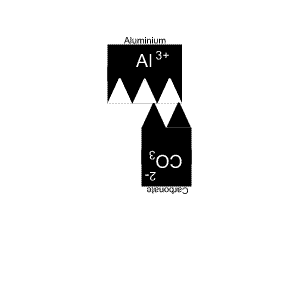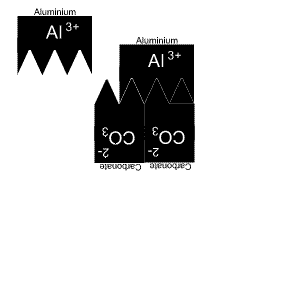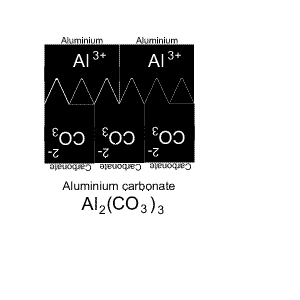
It takes two aluminium and three carbonate ions to complete the compound.
Now, if we need more than one ion we need to use subscripts.
We use brackets only if both of the conditions below exist:
-
the ion has more than one capital letter
and
- more than one ion is needed.
Aluminium has only one capital letter so we do not need brackets. But carbonate has two capital letters so we need brackets.
Chloride(Cl- ) and aluminium (Al3+)
Solution

Carbonate (SO42-) and aluminium (Al3+)
Solution