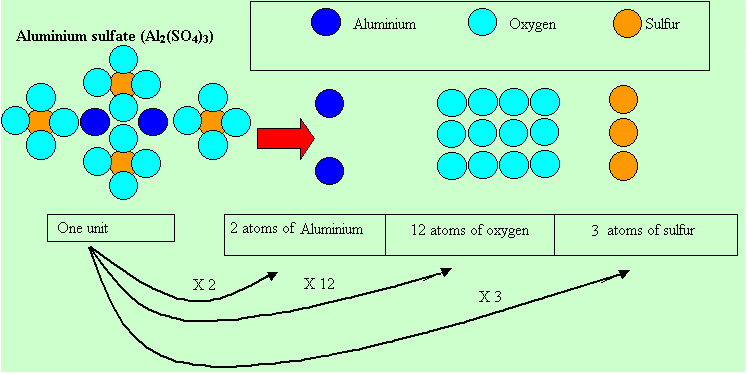

One mole of
![]()
units(![]() )
)
contains three times as many sulfur atoms.
So
3 X ![]() =
=![]()
atoms of sulfur.
3.42 grams of
![]()
must be converted to moles first,
(divide by its formula mass (342 atomic mass units)).
3.42 grams/342 atomic mass units
= 0.01mole.
So! the number of mole of aluminium atoms is 3 X 0.01 =0.03mole.
So the number of atoms of aluminium is
0.03 X ![]() =
= ![]()
|
Mole exercises
Al = 27, S =32, O=16 |
||||||||||||||||||||||||||||||||||||
|