|
Monomers are small molecules that can link end to end to form long polymers.
This reaction is known as polymerisation.
Monomers have two or more sites where joining can occur.
Consider the monomers below.
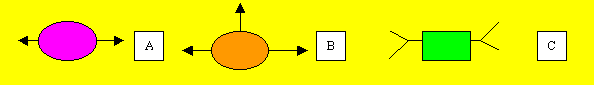
Consider the polymerisation
of "A" and "C"
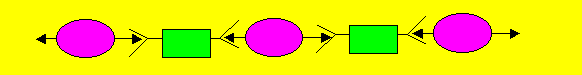
In this instance a linear(straight) polymer is formed. Linear polymers
pack together to form a thermoplastic. Thermoplastics are plastics that
melt when heated.
Consider the polymerisation of "B" and "C"
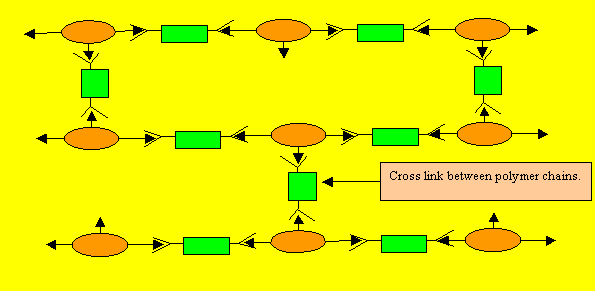
Notice how the
polymers are linked to one another. They link to one another like bricks
on a wall. Such polymers are known as thermosetting plastics. Thermosetting
plastics can not be softened with heat.
The choice of a monomer is crucial in producing the right polymer. Any
monomer with more than two joining sites can produce thermosetting plastics.
Thermosetting plastics are also known as cross-linked polymers.
|


