Carboxylic acids
Aldehydes
Ketones
Alcohols
Amines
Alkenes
Alkynes
Halides (bromo, chloro, iodo, etc)
Alkyls groups (methyl, ethyl, etc.)
| Functional Group | As Priority Group (Suffix) | As Substituent (Prefix) |
|---|---|---|
| OH (hydroxyl) |
-ol
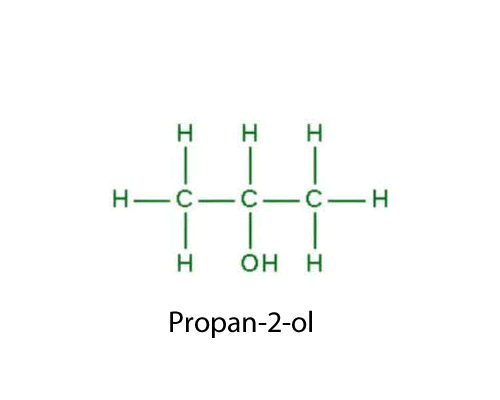
|
hydroxy- 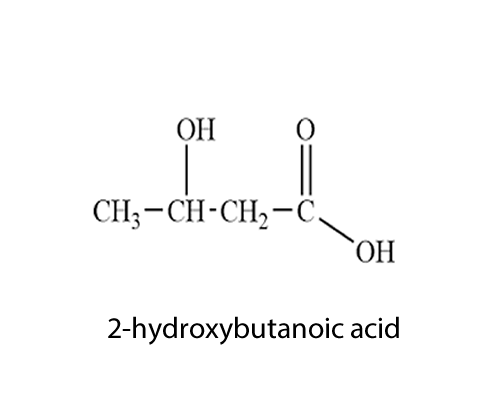
|
| NH2 (amino) |
-amine
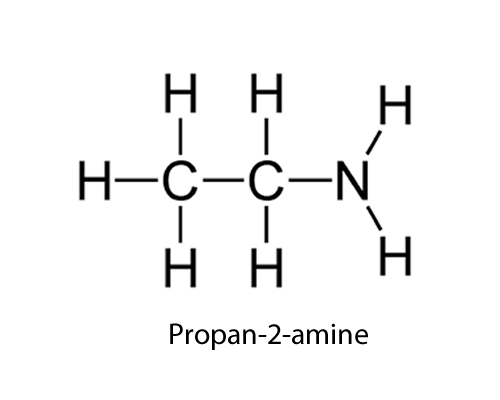
|
amino- 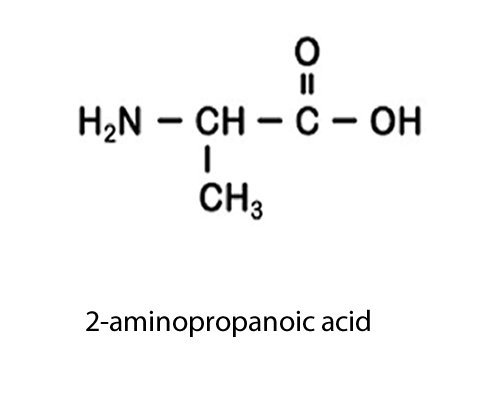
|
| CHO (aldehyde) |
-al
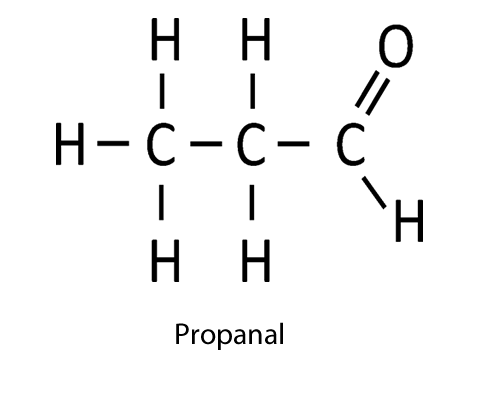
|
oxo- 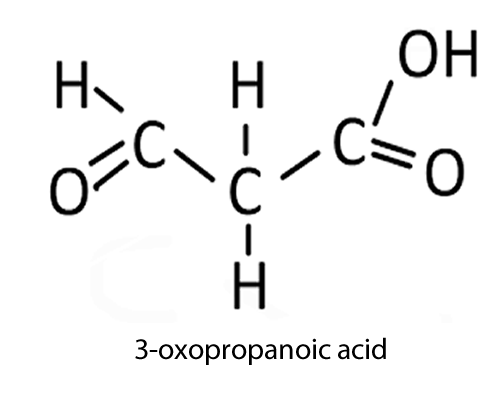
|
| C=O (ketone) |
-one
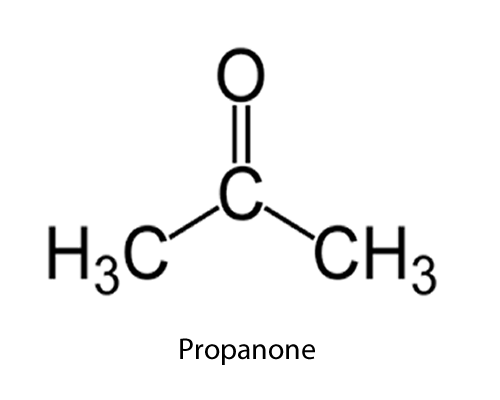
|
oxo-
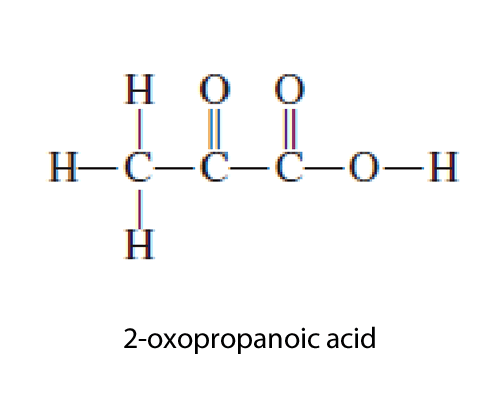
|
Now let's first of all look at amines and alcohols.
Alcohols have the hydroxy (OH) functional group while amines have the amino (NH2) functional group. The question here is when should you use the terms hydroxy or amino instead of alcohol and amine.
Simple. If the hydroxy functional group is the priority then the molecule is an alcohol and has a suffix of ol, but if its not the priority it has the prefix of hydroxy. If the amino functional group is the priority then we use the suffix amine, however, if it is not a priority we use the prefix amino.
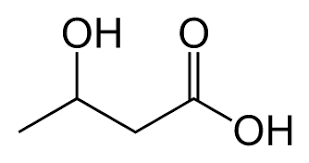
Consider the molecule on the right. The carboxyl functional group is the priority. The molecule is clearly an acid where the carbon backbone is butanoic acid. Coming off the third carbon is a hydroxyl group. So the name of this molecule has the prefix hydroxy and is given the name 3-hydroxybutanoic acid.
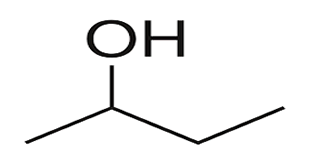
So the name of this molecule is butan-2-ol
Lets see how this is applied.
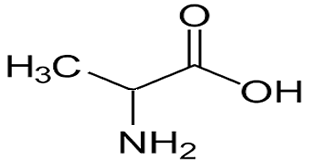
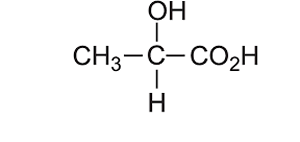
Consider the compound shown on the right.
it consists of two functional groups, hydroxy and a carboxyl group. Is it an alcohol or is it an acid? According to the priority list the root name is a carboxylic acid.
So it is propanoic acid with a hydroxy group on carbon number two.
So it is 2-hydroxypropanoic acid.
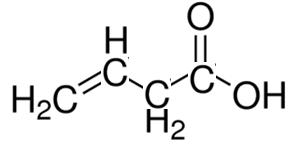
Consider the compound shown on the right.
It consists of two functional groups, carboxyl and a double C=C bond.
According to the priority the root chain of carbons must include the carboxyl group. So it is a carboxylic acid with a double bond on carbon number three.
*** Note that since the parent molecule is an acid and not an alkene the last "e" in the ene is removed.
The compound is 3-butenoic acid
or but-3-enoic acid
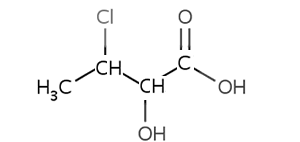
Consider the compound shown on the right.
It consists of three functional groups, carboxyl, hydroxy and chloro.
According to the priority the root chain of carbons must include the carboxyl group. So it is a carboxylic acid with a hydroxy group on carbon number 2 and a chloro on carbon number 3.
The compound is 3-chloro-2-hydroxybutanoic acid
Consider the compound shown on the right.
It consists of two functional groups, hydroxy and an amino. According to the priority it is an alcohol with an amino group on carbon number 3.
The compound is 1-amino-2-propanol
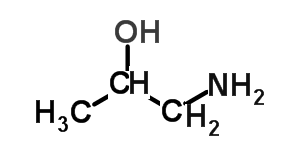
Consider the compound shown on the right.
It consists of two functional groups, carboxyl and an amino. According to the priority it is a carboxylic acid with an amino group on carbon number 3.
The compound is 3-aminobutanoic acid
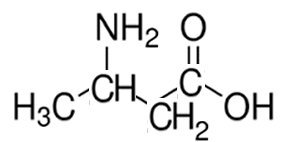
Consider the compound shown on the right.
It consists of three functional groups, a carbon to carbon double bond a chloro and a hydroxy. According to the priority it is an alcohol with a chloro group on carbon number 3 and a double bond on carbon number 2.
The compound is 3-chloro-2-buten-1-ol or 3-chlorobut-2-en-1-ol.
Notice how the last e in butene is removed and the suffix -1-ol added.
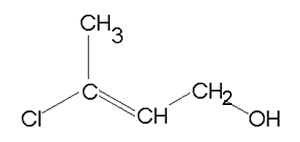
Consider the compound shown on the right.
It consists of three functional groups, a carbon to carbon double bond a chloro and a methyl. According to the priority it is an alkene with a chloro group on carbon number 1 and a methyl group on carbon number 2.
The compound is 1-chloro-2-methyl-2-butene
Solution
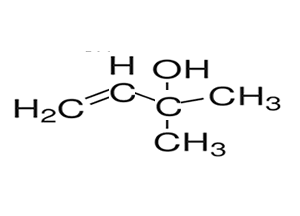
2) Name the compound on the right
Solution
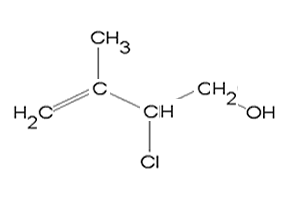
3) Name the compound on the right
Solution
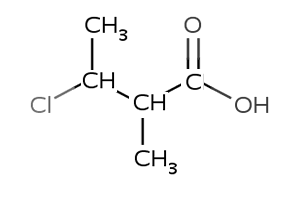
4) Name the compound on the right
Solution
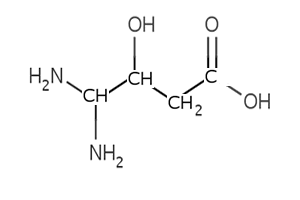
5) Name the compound on the right
Solution
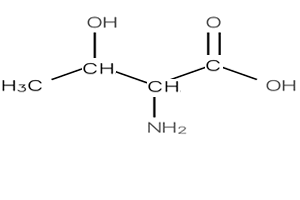
6) Name the compound on the right
Solution
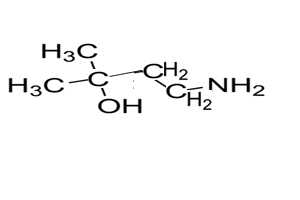
7) Name the compound on the right
Solution
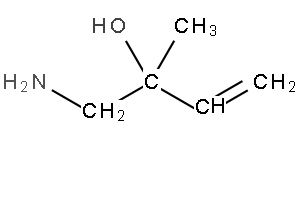
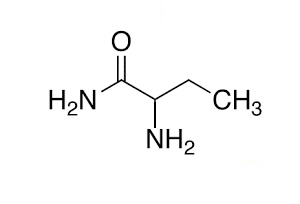
8) Name the compound on the right (*note that naming amides is not on the 2023-2027 VCE study design)
Solution
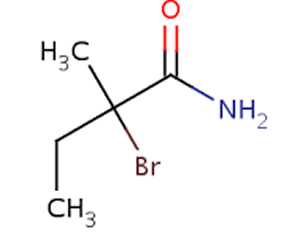
9) Name the compound on the right (*note that naming amides is not on the 2023-2027 VCE study design)
Solution
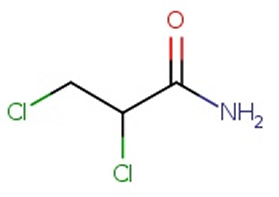
10) Name the compound on the right. (*note that naming amides is not on the 2023-2027 VCE study design)
Solution