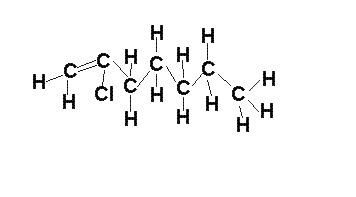
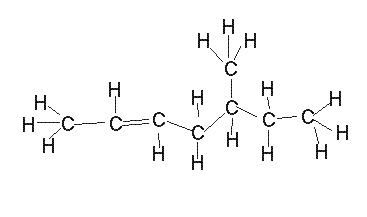
The alkenes
When dealing with molecules which have a double carbon to carbon bond follow the following steps.
1) Determine the longest chain of carbon atoms that includes the double bond.
2) Name the carbon atoms so that the double bond is on the lowest carbon.
3) Each of the substituents and the number of the carbon to which they are attached are placed in the name as prefixes in alphabetical order, followed by the longest
carbon chain and finally the location of the double bond.
Eg 5-methylhept-3-ene has a seven carbon backbone with a double bond at carbon number 3 and a methyl group on carbon number 5.
**notice how in the suffix of the name the "ane" of heptane is replaced with "ene" for heptene.
Let's see an example.
In the presence of multiple double bonds, however, the following steps are taken:
-
number of double bonds is indicated by the appropriate suffix such as -diene or -triene. Numbers before the suffix give the carbon number that is relevant to the location of each carbon to carbon double bond on the backbone or parent carbon chain.
-
only -ne is removed from the parent alkane chain name leaving an "a" in the name.
Let's see an example
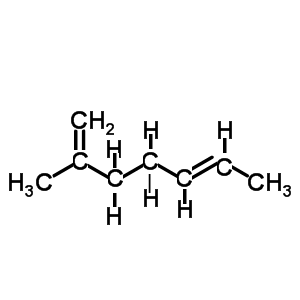
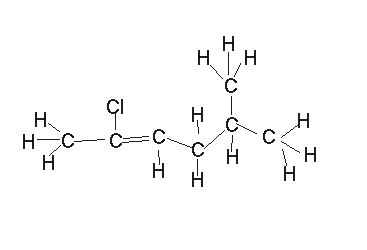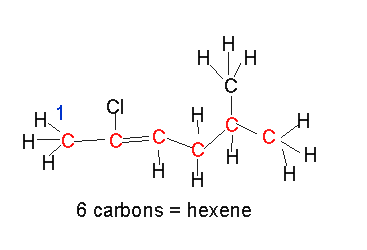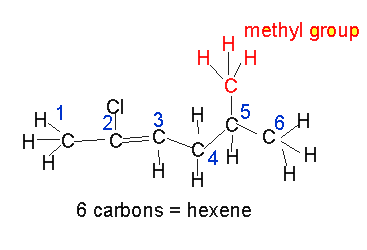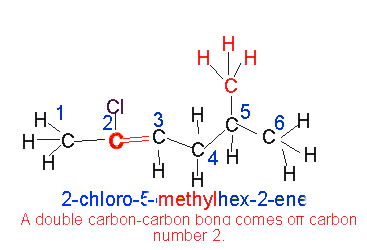
Name the diene shown on the left.
- select the longest chain that includes both C=C bonds.
It is a hepta-diene
- number the carbons so that the C=C are on the lowest possible numbered carbons.
hepta-1,5-diene
- identify the substituent groups and the carbon number to which they are attached and add it to the name as a prefix.
2-methylhepta-1,5-diene