Aspirin is a compound widely used as a painkiller and to relieve the symptoms of fever. It can be produced by
means of a reaction in which salicylic acid is one of the reagents.
The structures of aspirin and salicylic acid are shown below.
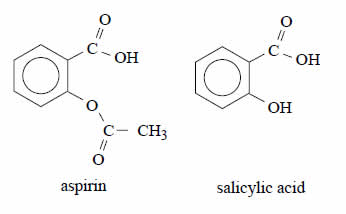
Which one of the following statements about aspirin is not correct?
A. Aspirin may be prepared by reaction between salicylic acid and
CH3OH.
B. Aspirin contains both an ester and a carboxylic acid functional group.
C. Aspirin can undergo an acid-base reaction with NaHCO3.
D. Aspirin may be prepared by reaction between salicylic acid and
CH3COOH.

Capsaicin is an important component of some pain relief ointments. It is also the major compound responsible for the burning sensation of chilli peppers.
A structure for capsaicin is given below.
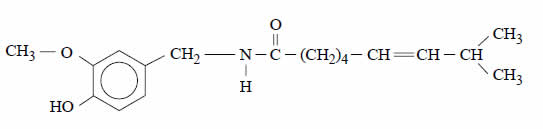
A molecule of capsaicin contains
A. an ester functional group and an amide functional group.
B. an ester functional group and an alcohol functional group.
C. an alkene functional group and an amide functional group.
D. a carboxylic acid functional group and an alcohol functional group.
Solution

A student was given the task of identifying a liquid organic compound that contains only carbon, hydrogen and
oxygen. The following tests were carried out

Based on the above test results, the compound could be
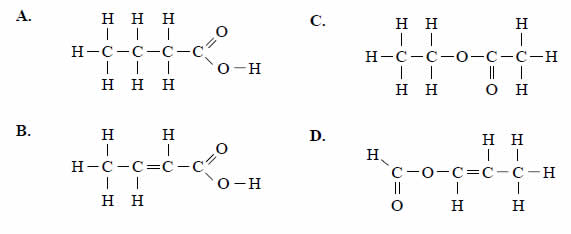
Solution

Starch consists mainly of amylose, which is a polymer made from glucose, C6H12O6. A particular form of
amylose has a molar mass 3.62 × 105 g mol–1.
A molecule of this amylose can be described as
A. an addition polymer of 2235 glucose molecules.
B. an addition polymer of 2011 glucose molecules.
C. a condensation polymer of 2235 glucose molecules.
D. a condensation polymer of 2011 glucose molecules.
Solution

In a laboratory experiment, a mixture of alkanes was separated into components by fractional distillation using
the following apparatus.
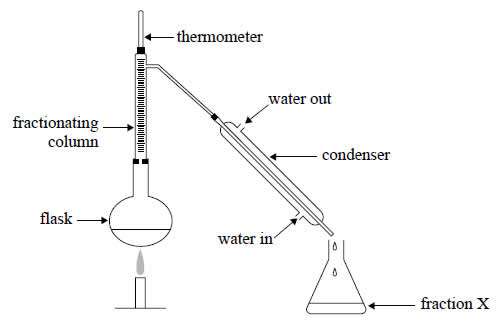
The fi rst fraction collected is fraction X, then fraction Y then fraction Z.
From this information we can deduce that
A. fraction Y is more volatile than Z.
B. fraction Y has a higher molar mass than Z.
C. fraction X has a higher boiling point than Y and Z.
D. fraction Z has stronger covalent bonds in its molecules than X and Y.
Solution

A mixture contains several different organic liquids all of which boil at temperatures greater than 50°C.
The compounds present in the mixture are separated. Three of the compounds, compounds X, Y and Z, are
analysed as follows.
Compound X is vaporised. At a temperature of 120°C and a pressure of 115 kPa, a 0.376 g sample of the vapour occupies 124 mL.
a. Calculate the molar mass, in g mol–1, of compound X.
Solution

Compound Y is an alkanol of molecular formula of C4H10O.
b. Draw the structural formulae, showing all bonds, of the four possible alkanols with a molecular formula of
C4H10O.
Solution

Compound Y shows 3 lines in the 13C NMR spectrum and undergoes reaction with Cr2O7 2– (aq) in acid to
produce a carboxylic acid.
c. What evidence about the structure of Y can be gained from this information?
Solution

d. Based on the above evidence, identify compound Y by writing its systematic name.
Solution

Give a systematic name for each of the following compounds.
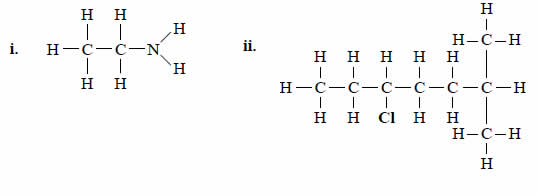
Solution

fuels.
Write a chemical equation for the combustion of ethanol in excess oxygen.
Solution
Ethanol is fully miscible with water. It is also fully miscible with petrol (a mixture of hydrocarbons).
Give chemical reasons why ethanol can mix with both water and petrol.
Solution
Ethanol can be produced by fermentation of glucose.
i. Write a chemical equation for the fermentation of glucose to produce ethanol.
Solution
ii. Explain why ethanol produced by fermentation is referred to as a ‘biochemical fuel’.
Solution
