Step 1 The longest chain of carbons takes the root name (alkanamine) and the other chains become a substituent with the letter N (italicised) to indicate it is bonded to the nitrogen.
Lets see some examples
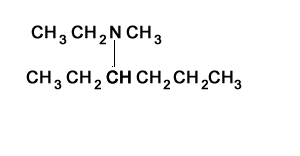
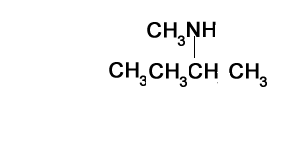
Consider the structure shown on the right. It represents a secondary amine as the nitrogen is bonded to two carbons.
Step 1) Identify the longest alkanamine
Step 2) Identify the substituent chains branching off the nitrogen
Step 3) Write the name using N to indicate the attachment to the nitrogen atom. Note the use of di.
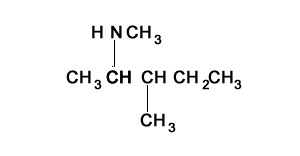
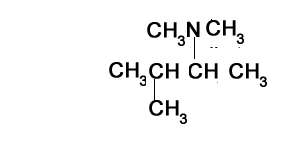
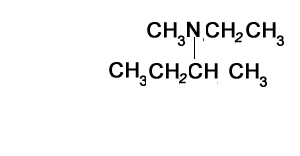
Consider the structure shown on the right. It represents a tertiary amine as the nitrogen is bonded to three carbons.
Step 1) Identify the longest alkanamine
Step 2) Identify the substituent chains branching off the nitrogen
Step 3) Write the name using N to indicate the attachment to the nitrogen atom. Notice how the ethyl and methyl groups are placed in alphabetical order when attached to the same carbon or nitrogen.
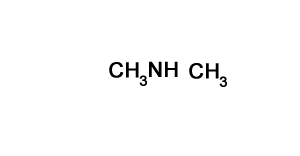
Solution
Name
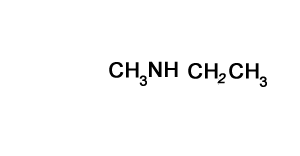
N-methylpropan-1-amine
Solution

N-ethyl-N-methylbutan-1-amine
Solution

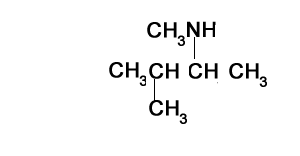
N,N-dimethylbutan-2-amine
Solution
