Solution
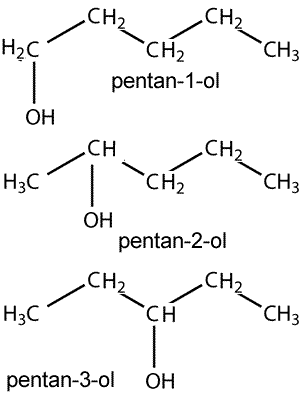
9) Consider the three alcohols shown on the right. Place them in order of increasing boiling point and give reasons.
There are three main factors that influence boiling points.
1) The intermolecular forces acting between molecules
2) Molecular mass
3) Molecular symmetry and branching.
All three molecules have the same functional group and so are capable of exhibiting some form of hydrogen bonding and have the same molecular mass. So it comes down to symmetry and branching.
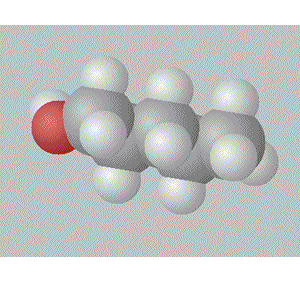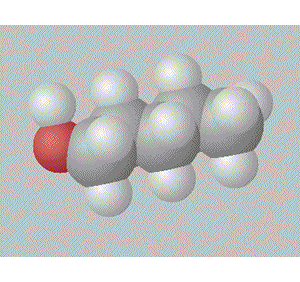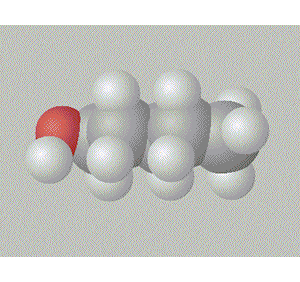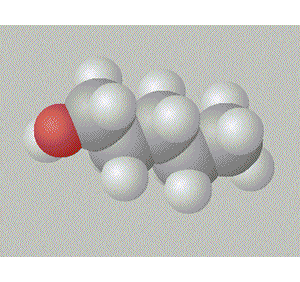
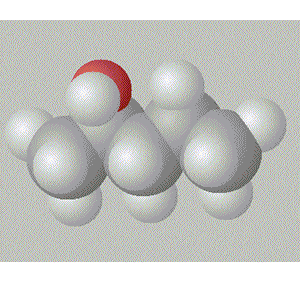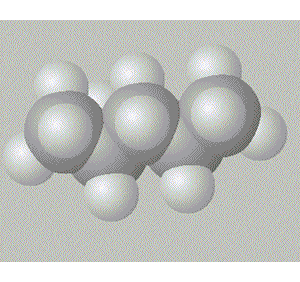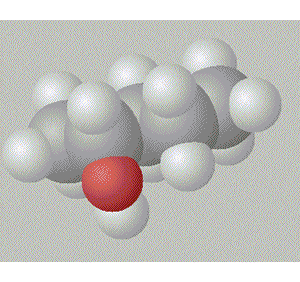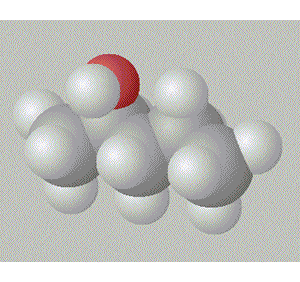
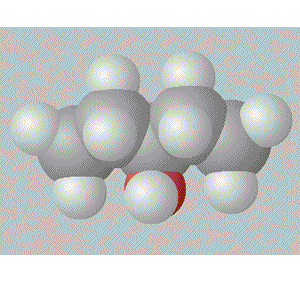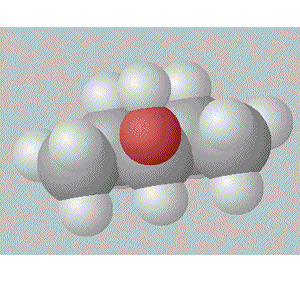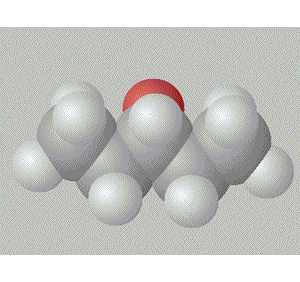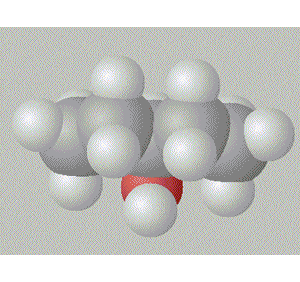
Pentan-1-ol is the most assymetrical, molecule and so is the most polar, hence, it will have the highest boiling point.
Pentan-2-ol has the hydroxy group in between a methyl and a propyl group so it is relatively surrounded by non-polar alkyl groups.
Pentan-3-ol has the hydroxy group in a relatively inaccesible place, surrounded by two ethyl groups right in the middle of the molecule. This also gives this molecule a degree of symmetry.
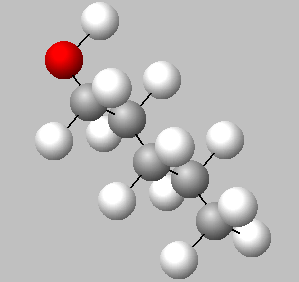
A space fill model of each molecule better shows how the hydroxy group is shielded in each molecule.