Particle theory
-difference between heat and temperature
Below are the properties of solids, liquids and gases.
| Property | Solid | Liquid | Gas |
| Shape and volume | Definite shape and volume | Definite volume but no definite shape. | Takes on the shape and volume of its container. |
| Ability to flow | no | yes | yes |
| Compression | very little | very little | yes |
| Effect of heat on volume. | little | little | great |
The particle theory states that:
-all matter is made of small particles( atoms or molecules);
-these particles are in constant motion and are held together by electrostatic forces.
The particles of a solid are held together in fixed positions by strong forces and can only vibrate, as shown on the right. As the solid substance becomes hotter its particles vibrate more violently.
Click to see the particles of a solid. Now Click to see the same particles when heated to a very high temperature. What can you say about the volume of the solid?

The particles of a liquid are also held together by strong forces but the particles have enough energy to momentarily break free from these forces and so move about, always maintaining some kind of contact with each other.
When liquid particles are heated they also move faster and move further apart. This increases slightly the volume of the mass of liquid being heated.
The particles of a gas have enough energy to break free from any forces of attraction and move about at high speed traveling in straight lines and colliding with the wall of the container and occasionally each other. Click to see the gas at a higher temperature.
Explain the difference you observe.

What is the difference between heat and temperature? Often these two terms are confused. Heat is the amount of energy given to a particular substance but temperature is a measure of the average kinetic energy of its particles.
Say 1 gram of water and 1 gram of oil were each given 100 joules of heat energy. The temperature of the oil would increase more than the temperature of the water. From this we know that the average kinetic energy of the oil molecules is higher than the average kinetic energy of the water molecules. The oil molecules are moving around with a higher average speed than the water molecules.
The particles of the solid substance, shown on the right, are at a higher temperature than the particles of the solid substance shown above. Why?
Which comment is true?
When heated ;


Which stage represents the hot solid state? Explain why.
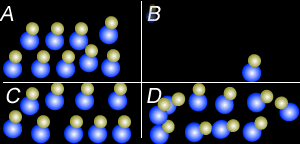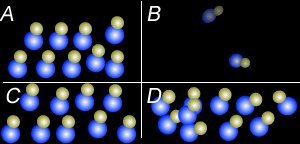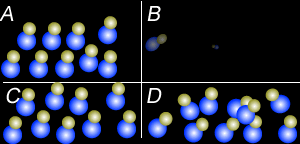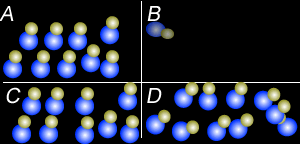
Using the particle theory explain why:
- Liquids and solids can not be compressed to any great extent but gases can.
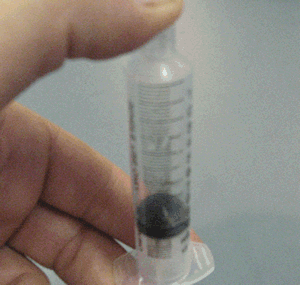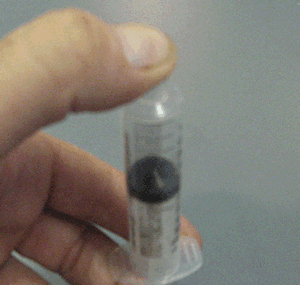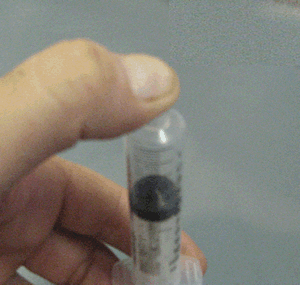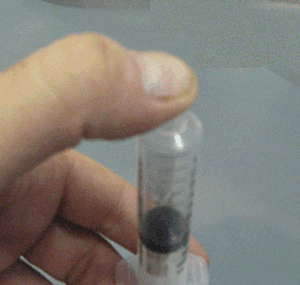
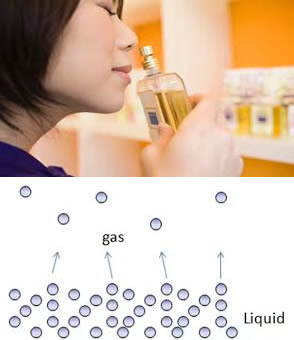
- Why when a bottle of perfume is opened it can be smelt from across the room quickly.
- Why is the smell of perfume stronger on a hot day than on a cold day.
One gram of both substance "A" and "B" are given 100 joules of heat energy. The temperature of substance "A" rises by
1 oC while those of substance "B" rise by 10 oC.
Describe the difference between the particles of each substance?
During a bush-fire burning wood can form a dangerous thick blanket of wood particles in the gas state. We generally call this smoke. Smoke, however, is made of unburnt wood particles and is as flammable as petrol.
Petrol burns easily because its particles readily form a thick layer of gas on the surface that can quickly catch fire if a naked flame comes into contact with the gas. This is the reason why no smoking signs are everywhere in petrol stations.
Consider the two videos on the right.
Warning. Do not try this at home without adult supervision.
Offer some safety advise to firefighters when fighting bush-fires with reference to particle theory.
A drop of red dye is placed in each beaker of water, as shown in the time lapse image on the right.
a) Which beaker has the hottest temperature? Explain.
b) Using the particle theory offer an explanation as to why the dye disperses slowly in beaker B than in beaker A.

Cart wheels were the main used for many years to maintain horse drawn carts transporting cargo and people. People used knowledge of particle theory to make sturdy tyres, made of steel, that wrapped around wooden wheels.
Step 1 Initially the metal tyre is heated.
Using particle theory explain why the metal tyre has to be heated?.
Step 2 The hot tyre is then hammered into place around the frame of the wooden wheel.
Step 3. The tyre is then sprayed with cold water.
How does cooling with water assist the tyre to grip tightly onto the wooden frame?
