For quality control, a chemist analyses the vitamin C (molecular formula C6H8O6) content of a new brand of fruit juice. The reaction used is an oxidation-reduction reaction in which I3- is the oxidant and vitamin C is the
reductant.
The reaction is
C6H8O6(aq) + I3- (aq) →C6H6O6(aq) + 2H+(aq) + 3I-(aq)
The half reaction for the oxidation of I-(aq) to I3- (aq) is
I3- (aq) + 2e → 3I-(aq)
Give the half reaction for the oxidation of vitamin C.
Solution
A 20.00 mL sample of the fruit juice was made up to 250.0 mL with pure water in a volumetric flask.
25.00 mL aliquots of the diluted fruit juice were then placed in a conical flask and titrated with a solution
in which [I3-] = 2.00 X 10-4 M.
An average titre of 15.65 mL was obtained.
Calculate the amount of I3- present in the average titre, in mole.
Solution
Calculate the amount of vitamin C present in each 25.00 mL aliquot, in mole.
Solution
Calculate the concentration of vitamin C in the original (undiluted) sample of fruit juice in mole per
litre.
Solution

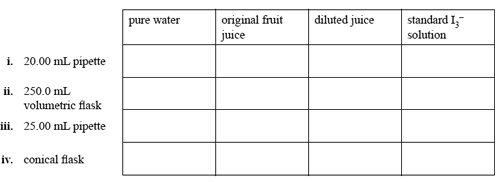
During the analysis the chemist rinsed, but did not dry, each item of glassware. She had available for use:
pure water, the original fruit juice, the diluted fruit juice and the standard
I3- solution. For each item listed below, name the liquid that should be used to rinse it by placing a tick in the appropriate box.
Solution
