A chemical reaction has a ΔH of -150 kJ mol- and the activation energy for its reverse reaction is 350 kJ mol-.
The activation energy, in kJ mol-, of the forward reaction is
A. +500
B. +200
C. +150
D. -200
Solution

The reaction
A + B → C; ΔH negative
involves a two-step process
A + B → X; ΔH positive
X → C; ΔH negative
Which one of the following diagrams best represents the energy changes during the course of the reaction?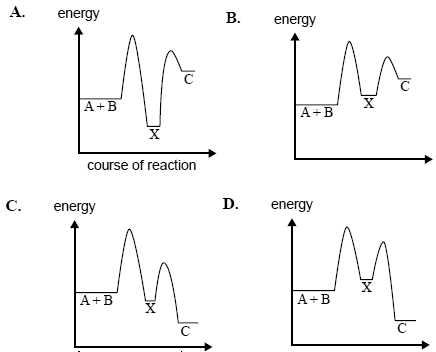
Solution
