Many industrial processes create waste products such as chimney gases. These gases may contain serious atmospheric
pollutants, such as oxides of nitrogen (for example, NO and NO2).
One way to remove these nitrogen oxides is to treat the chimney gases with ammonia. This treatment converts the oxides
of nitrogen in the chimney gases to nitrogen and water. These are then released into the atmosphere.
a. i. Determine the coefficients that correctly balance the equation for this reaction. Write your answers in the
spaces provided.
NO(g) + NH3(g) → N2(g) + H2O(g)
Solution
It is important to adjust the amount of ammonia mixing with the chimney gases to give the correct mole ratio of ammonia to nitrogen(II) oxide, NO.
ii. Explain the effect on the composition of the gases released into the atmosphere if the amount of ammonia
was
- too low ______________________________________
- too high ______________________________________
Solution
b. The ammonia can be produced on-site in industrial plants using small-scale ammonia generators. The ammonia is produced by reacting urea with water. A simplified diagram of such a plant is provided below.
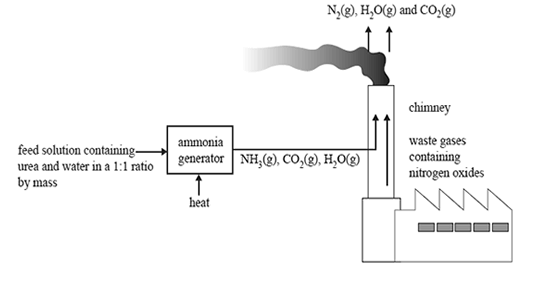
The chemical reaction occurring in the ammonia generator is
(NH2)2CO(aq) + H2O(l) => 2NH3(aq) + CO2(aq) ΔH + ve
In a particular generator a 1:1 mass ratio of urea and water is used.
i. Which reactant is in excess?
Solution

c) Give one reason why an excess of one reactant is used in this chemical reaction.
Solution
Changing the temperature of the reaction mixture in the ammonia generator can control the amount of ammonia gas produced.
d) Explain the effect of increasing the temperature on the amount of ammonia formed in the generator.
Solution
The table gives some of the properties of ammonia and urea.
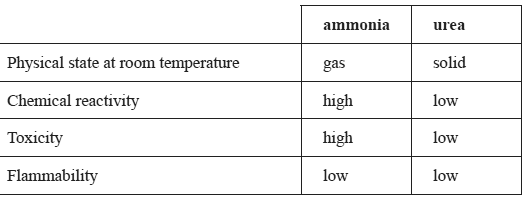
e) Give one advantage, other than cost, of producing ammonia on-site by this method rather than having large quantities of ammonia delivered from a plant at another location.
Solution
