Mass-spectrometry exercises (2010 VCE)
1) The molecules ethanol and nitrogen dioxide have the same molar mass (M = 46 g mol–1). They can be easily distinguished
by mass spectrometry.
The mass spectra of the two molecules are shown below.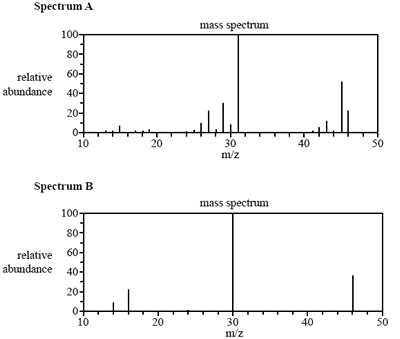
a. Write an equation showing how either an ethanol molecule or a nitrogen dioxide molecule becomes ionised in the mass spectrometer. (1 mark)
Solution
b. Which mass spectrum cannot be that of nitrogen dioxide? What evidence does the mass spectrum provide to support your answer? (2 marks)
Solution
c. What is the formula of the species that produces the peak seen at m/z 30 in spectrum B? (1 mark)
Solution
