Redox (2010 NSW HSC)
1)In which of the following reactions does the metal atom show the greatest change in
oxidation state?
(A) MnO4− to Mn2+
(B) MnO2 to Mn(OH)3
(C) PbO2 to PbSO4
(D) VO2+ to VO2+
Solution
2) Chromate and dichromate ions form an equilibrium according to the following equation.
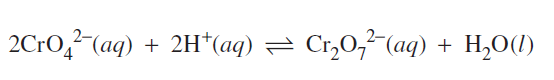
Which solution would increase the concentration of the chromate ion (CrO4-2) when added to the equilibrium mixture?
(A) Sodium nitrate
(B) Sodium chloride
(C) Sodium acetate
(D) Ammonium chloride
Solution
