Thermochemistry (2010 NSW HSC)
1)Write a balanced chemical equation for the complete combustion of 1-butanol.
Solution
(b) A student measured the heat of combustion of three different fuels. The results are shown in the table.
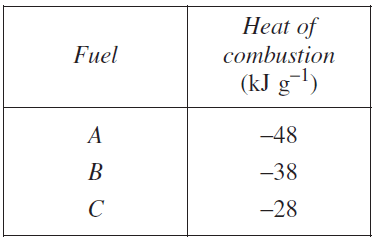
The published value for the heat of combustion of 1-butanol is 2676 kJ mol−1.
Which fuel from the table is likely to be 1-butanol? Justify your answer.
Solution
