1) The IR wavenumber for bond stretching in a C-O bond (1000-1300 cm-) is lower than for a C-H bond (2850-3300 cm-). Which one of the following statements best explains this fact?
a) Oxygen atoms are more electronegative than hydrogen atoms
b) Oxygen atoms have a greater atomic mass than hydrogen atoms.
c) Oxygen atoms have a greater atomic radius than hydrogen atoms.
d) Oxygen atoms have a higher nuclear charge than hydrogen atoms..
Solution

2) Petrol is a mixture of hydrocarbon molecules varying in size from six to ten carbon atoms. Forensic investigators suspect that traces of a substance found at a suspicious fire could be petrol that was used to start the fire. Which one of the following techniques could best be used to identify the substance?
a) NMR spectroscopy
b) UV-Visible spectroscopy
c) atomic absorption spectroscopy
d) gas chromatography followed by mass spectroscopy.
Solution

3) The amount of copper in a solution of copper(II) sulfate can be determined using atomic absorption spectroscopy. When a blue copper (II) sulfate solution is introduced into an atomic absorption spectrometer, a green flame is observed. Consider the following.
I) A copper (II) sulfate solution appears blue because it absorbs red light.
II) The metal species undergoes oxidation in the flame.
III) The flame is green due to electron transitions from a higher energy state to a lower energy state.
Which of the above statement are true?
a) ! only
b) I and III only
c) II and III only
d) I, II and III
Solution

4)
Bromine exists as two isotopes,79Br and 81Br. The mass spectrum of bromoethane, C2H5Br with two molecular ion peaks at m/z 108 and 110, is shown below.
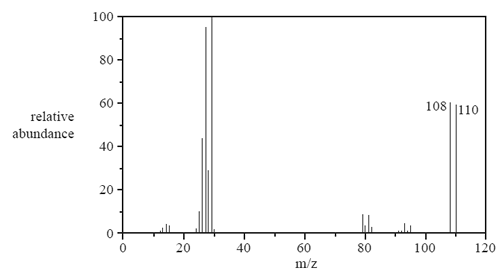
a) Identify the species that produces the peak at m/z = 29.
Solution
d) What do the two molecular ion peaks indicate about the relative abundance of 79Br and 81Br? Give a reason
Solution

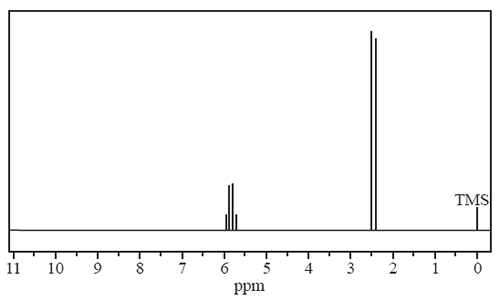
i) Draw the structural formula of each of the two compounds with the molecular formula C2H4Br2.
Solution
ii) Which one of your two structures above corresponds to the 1H NMR spectrum provided? Justify your answer by referring to both the 1H NMR spectrum and to the structure of the compound.
Solution
