1) Fuel cells have a number of applications that offer advantages over conventional methods of electricity generation.
Which one of the following is not a feature of modern fuel cells?
A. They generate very little noise.
B. They are a cheap source of electricity.
C. They enable electricity to be generated on site.
D. They have the potential to reduce emissions of carbon dioxide into the atmosphere.
Solution


2) Methanol is suitable for use in a micro fuel cell that is used to power laptop computers and similar small electrical items. The methanol is oxidised to carbon dioxide and water. The half-equation for the anode reaction is
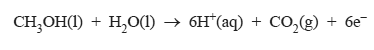
i. Write a balanced half-equation for the cathode reaction.
Solution
ii) A finely divided platinum/ruthenium catalyst is used in this cell. Give a reason why it is important to have a catalyst that will signifi cantly reduce the activation energy for
the cell reaction.
Solution
