![]()
1) a)

Solution

b) A concentration–time graph for this system is shown below.
concentration
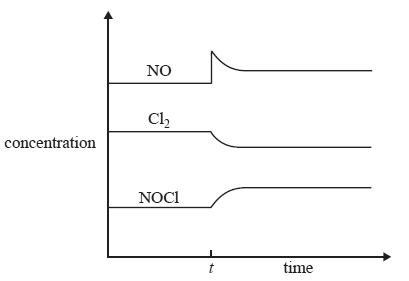
What event occurred at time t to cause the change in equilibrium concentrations?
A. The pressure was decreased at a constant temperature.
B. The temperature was increased at a constant volume.
C. A catalyst was added at a constant temperature and volume.
D. Additional NO gas was added at a constant volume and temperature
Solution
2) The industrial production of hydrogen involves the following two reactions.
![]()
a) Write 'increase', 'decrease' or 'no change' in the table below to identify the expected effect of each change to reaction I and reaction II on the equilibrium yield of hydrogen.
| Change to reaction I and reaction II | Effect of the change on the hydrogen yield in reaction I | Effect of the change on the hydrogen yield in reaction II |
| addition of steam at a constant volume and temperature |
||
| increase in temperature at a constant volume |
||
| addition of a suitable catalyst at a constant volume and temperature |
Solution

b) Explain the effect of decreasing the volume, at constant temperature, on the hydrogen equilibrium yield in each reaction.